Soluble transferrin receptor as a marker of erythropoiesis in patients undergoing high-flux hemodialysis
DOI:
https://doi.org/10.17305/bjbms.2017.1972Keywords:
Hemodialysis, renal anemia, high-flux dialysis, soluble transferrin receptor, HFD, erythropoiesis, sTfR, iron statusAbstract
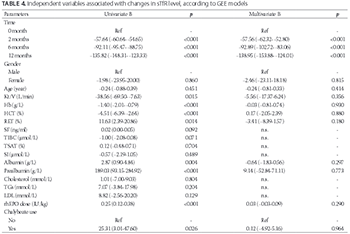
Anemia is a common complication in chronic kidney disease (CKD) patients receiving hemodialysis. The effect of high-flux dialysis (HFD) on anemia remains unclear. This prospective study aimed to evaluate the effect of HFD on anemia, and the potential of soluble transferrin receptor (sTfR) as a marker of iron status and erythropoiesis in CKD patients on hemodialysis. Forty patients, who switched from conventional low-flux dialysis to HFD for 12 months, were enrolled in this study. The levels of sTfR, hemoglobin (Hb), iron, and nutritional markers, as well as the dose of recombinant human erythropoietin (rhEPO) and use of chalybeate were determined at 0, 2, 6, and 12 months after starting HFD. HFD significantly increased the hemoglobin level and reduced sTfR level in CKD patients (p < 0.05). In addition, significant decreasing linear trends were observed for rhEPO dosage and chalybeate use (p < 0.05). The level of sTfR was positively correlated with the percentage of reticulocytes (RET%), rhEPO dose, and chalybeate use, while it was negatively correlated with Hb levels and total iron-binding capacity results (all p < 0.05). A univariate generalized estimating equation (GEE) model showed that the Hb level, RET%, rhEPO dose, and chalybeate use were the variables associated with sTfR levels. A multivariate GEE model showed that the time points when hemodialysis was performed were the variables associated significantly with sTfR levels. Overall, our findings suggest that HFD can effectively improve renal anemia in hemodialysis patients, and sTfR could be used as a marker of erythropoiesis in HFD patients.
Citations
Downloads
References
Locatelli F, Bárány P, Covic A, De Francisco A, Del Vecchio L, Goldsmith D, et al. Kidney disease: Improving global outcomes guidelines on anaemia management in chronic kidney disease: A European Renal Best Practice position statement. Nephrol Dial Transplant 2013;28(6):1346-59. https://doi.org/10.1093/ndt/gft033.
Locatelli F, Aljama P, Canaud B, Covic A, De Francisco A, Macdougall IC, et al. Target haemoglobin to aim for with erythropoiesis-stimulating agents: A position statement by ERBP following publication of the trial to reduce cardiovascular events with Aranesp therapy (TREAT) study. Nephrol Dial Transplant 2010;25(9):2846-50. https://doi.org/10.1093/ndt/gfq336.
Ofsthun N, Labrecque J, Lacson E, Keen M, Lazarus JM. The effects of higher hemoglobin levels on mortality and hospitalization in hemodialysis patients. Kidney Int 2003;63(5):1908-14. https://doi.org/10.1046/j.1523-1755.2003.00937.x.
Babitt JL, Lin HY. Mechanisms of anemia in CKD. J Am Soc Nephrol 2012;23(10):1631-4. https://doi.org/10.1681/ASN.2011111078.
Zadrazil J, Horak P. Pathophysiology of anemia in chronic kidney diseases: A review. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 2015;159(2):197-202. DOI: 10.5507/bp.2013.093.
Tsagalis G. Renal anemia: A nephrologist’s view. Hippokratia 2011;15(Suppl 1):39-43.
Merello Godino JI, Rentero R, Orlandini G, Marcelli D, Ronco C. Results from EuCliD (European Clinical Dialysis Database): Impact of shifting treatment modality. Int J Artif Organs 2002;25(11):1049-60.
Ayli D, Ayli M, Azak A, Yüksel C, Kosmaz GP, Atilgan G, et al. The effect of high-flux hemodialysis on renal anemia. J Nephrol 2004;17(5):701-6.
Locatelli F, Andrulli S, Pecchini F, Pedrini L, Agliata S, Lucchi L, et al. Effect of high-flux dialysis on the anaemia of haemodialysis patients. Nephrol Dial Transplant 2000;15(9):1399-409. https://doi.org/10.1093/ndt/15.9.1399.
Schneider A, Drechsler C, Krane V, Krieter DH, Scharnagl H, Schneider MP, et al. The effect of high-flux hemodialysis on hemoglobin concentrations in patients with CKD: Results of the MINOXIS study. Clin J Am Soc Nephrol 2012;7(1):52-9. https://doi.org/10.2215/CJN.02710311.
Kliger AS, Foley RN, Goldfarb DS, Goldstein SL, Johansen K, Singh A, et al. KDOQI US commentary on the 2012 KDIGO Clinical Practice Guideline for Anemia in CKD. Am J Kidney Dis 2013;62(5):849-59. https://doi.org/10.1053/j.ajkd.2013.06.008.
Cameron JS. European best practice guidelines for the management of anaemia in patients with chronic renal failure. Nephrol Dial Transplant 1999;14(Suppl 2):61-5. https://doi.org/10.1093/ndt/14.suppl_2.61.
van Zyl-Smit R. Diagnosis and management of iron deficiency in chronic dialysis patients. Curr Opin Nephrol Hypertens 2000;9(6):669-74.
https://doi.org/10.1097/00041552-200011000-00013.
IV. NKF-K/DOQI clinical practice guidelines for anemia of chronic kidney disease: Update 2000. Am J Kidney Dis 2001;37(1 Suppl 1):S182-238. https://doi.org/10.1016/S0272-6386(01)70008-X.
Weiss G, Goodnough LT. Anemia of chronic disease. N Engl J Med 2005;352(10):1011-23. https://doi.org/10.1056/NEJMra041809.
Tarng DC, Hung SC, Huang TP. Effect of intravenous ascorbic acid medication on serum levels of soluble transferrin receptor in hemodialysis patients. J Am Soc Nephrol 2004;15(9):2486-93. https://doi.org/10.1097/01.ASN.0000137884.59308.E7.
Tarng DC, Huang TP. Determinants of circulating soluble transferrin receptor level in chronic haemodialysis patients. Nephrol Dial Transplant 2002;17(6):1063-9. https://doi.org/10.1093/ndt/17.6.1063.
Braga F, Infusino I, Dolci A, Panteghini M. Soluble transferrin receptor in complicated anemia. Clin Chim Acta 2014;431:143-7. https://doi.org/10.1016/j.cca.2014.02.005.
Chiang WC, Tsai TJ, Chen YM, Lin SL, Hsieh BS. Serum soluble transferrin receptor reflects erythropoiesis but not iron availability in erythropoietin-treated chronic hemodialysis patients. Clin Nephrol 2002;58(5):363-9. https://doi.org/10.5414/CNP58363.
Lorenzo JD, Rodríguez MM, Martín SS, Romo JM. Assessment of erythropoiesis activity during hemodialysis therapy by soluble transferrin receptor levels and ferrokinetic measurements. Am J Kidney Dis 2001;37(3):550-6. https://doi.org/10.1053/ajkd.2001.22079.
World Health Organization. Iron deficiency anaemia: Assessment, prevention, and control: A guide for programme managers. Geneva, Switzerland: World Health Organization; 2001.
Tonbul HZ, Kaya H, Selçuk NY, Tekin SB, San A, Akçay F, et al. The importance of serum transferrin receptor level in the diagnosis of functional iron deficiency due to recombinant human erythropoietin treatment in haemodialysis patients. Int Urol Nephrol 1998;30(5):645-51. https://doi.org/10.1007/BF02550560.
Gupta S, Uppal B, Pawar B. Is soluble transferrin receptor a good marker of iron deficiency anemia in chronic kidney disease patients? Indian J Nephrol 2009;19(3):96-100. https://doi.org/10.4103/0971-4065.57105.
Yoon SH, Kim DS, Yu ST, Shin SR, Choi du Y. The usefulness of soluble transferrin receptor in the diagnosis and treatment of iron deficiency anemia in children. Korean J Pediatr 2015;58(1):15-9. https://doi.org/10.3345/kjp.2015.58.1.15.
Choi JW, Pai SH. Reticulocyte subpopulations and reticulocyte maturity index (RMI) rise as body iron status falls. Am J Hematol 2001;67(2):130-5. https://doi.org/10.1002/ajh.1091.
Eleftheriadis T, Kartsios C, Liakopoulos V, Antoniadi G, Ditsa M, Papadopoulos C, et al. Does hepcidin affect erythropoiesis in hemodialysis patients? Acta Haematol 2006;116(4):238-44. https://doi.org/10.1159/000095873.
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-04-16
Published 2017-11-20









