NOD2/CARD15 gene mutations in patients with gouty arthritis
DOI:
https://doi.org/10.17305/bjbms.2016.1339Keywords:
Gouty arthritis, nucleotide binding and oligomerization domains/caspase recruitment domain-containing protein 15 gene mutations, prevalenceAbstract
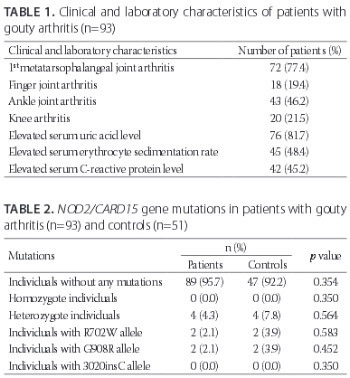
Nucleotide binding and oligomerization domains/caspase recruitment domain-containing protein 15 (NOD2/CARD15) is a cytoplasmic molecule controlling apoptosis and inflammatory processes by recognizing some microbial components. We aimed to identify the frequencies of NOD2/CARD15 gene mutations in patients with gouty arthritis and to determine their possible correlation with the disease phenotype. The study included 93 patients with gouty arthritis and 51 healthy controls matched for age, gender, and ethnicity. The NOD2/CARD15 R702W and G908R gene mutations were explored by the polymerase chain reaction restriction fragment length polymorphism method while the 3020insC mutation was analyzed by DNA sequencing. The mean patient age was 54.2 ± 14.2 years and mean duration of the disease was 3.1 ± 2.9 years. The first metatarsophalangeal and finger joint involvements were detected in 72 (77.4%) and 18 (19.5%) patients, respectively. Ankle arthritis and knee arthritis were detected in 43 (46.2%) and 20 (21.5%) patients, respectively. In total, 4 (9%) heterozygous mutations were detected in the G908R and R702W genes, while no mutation was detected in the 3020insC gene. Compared to the control group, there were no significant differences in all three DNA regions (G908R, R702W, and 3020insC; p = 0.452, p = 0.583, and p = 0.350, respectively). No correlation between the NOD2/CARD15 variants and clinical or laboratory findings (p > 0.05) was found. The frequencies of the NOD2/CARD15 gene mutations in the patients were similar to healthy control group. No association between clinical or laboratory findings and the NOD2/CARD15 gene mutations was observed.
Citations
Downloads
References
. Terkeltaub RA. Clinical practice. Gout. N Engl J Med 2003;349(17):1647-55. http://dx.doi.org/10.1056/NEJMcp030733.
. Arromdee E, Michet CJ, Crowson CS, O'Fallon WM, Gabriel SE. Epidemiology of gout: Is the incidence rising? J Rheumatol 2002;29(11):2403-6.
. Perez-Ruiz F, Castillo E, Chinchilla SP, Herrero-Beites AM. Clinical manifestations and diagnosis of gout. Rheum Dis Clin North Am 2014;40(2):193-206. http://dx.doi.org/10.1016/j.rdc.2014.01.003.
. Pope RM, Tschopp J. The role of interleukin-1 and the inflammasome in gout: Implications for therapy. Arthritis Rheum 2007;56(10):3183-8. http://dx.doi.org/10.1002/art.22938.
. Gutierrez O, Pipaon C, Inohara N, Fontalba A, Ogura Y, Prosper F, et al. Induction of Nod2 in myelomonocytic and intestinal epithelial cells via nuclear factor-kappa B activation. J Biol Chem 2002;277(44):41701-5. http://dx.doi.org/10.1074/jbc.M206473200.
. Hugot JP, Laurent-Puig P, Gower-Rousseau C, Olson JM, Lee JC, Beaugerie L, et al. Mapping of a susceptibility locus for Crohn's disease on chromosome 16. Nature 1996;379(6568):821-3. http://dx.doi.org/10.1038/379821a0.
. Caso F, Costa L, Rigante D, Vitale A, Cimaz R, Lucherini OM, et al. Caveats and truths in genetic, clinical, autoimmune and autoinflammatory issues in Blau syndrome and early onset sarcoidosis. Autoimmun Rev 2014;13(12):1220-9. http://dx.doi.org/10.1016/j.autrev.2014.08.010.
. Hugot JP, Chamaillard M, Zouali H, Lesage S, Cézard JP, Belaiche J, et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature 2001;411(6837):599-603. http://dx.doi.org/10.1038/35079107.
. Yao Q, Zhou L, Cusumano P, Bose N, Piliang M, Jayakar B, et al. A new category of autoinflammatory disease associated with NOD2 gene mutations. Arthritis Res Ther 2011;13(5):R148. http://dx.doi.org/10.1186/ar3462.
. Bonen DK, Ogura Y, Nicolae DL, Inohara N, Saab L, Tanabe T, et al. Crohn's disease-associated NOD2 variants share a signaling defect in response to lipopolysaccharide and peptidoglycan. Gastroenterology 2003;124(1):140-6. http://dx.doi.org/10.1053/gast.2003.50019.
. Arnott ID, Nimmo ER, Drummond HE, Fennell J, Smith BR, MacKinlay E, et al. NOD2/CARD15, TLR4 and CD14 mutations in Scottish and Irish Crohn's disease patients: Evidence for genetic heterogeneity within Europe? Genes Immun 2004;5(5):417-25. http://dx.doi.org/10.1038/sj.gene.6364111.
. Kim TH, Rahman P, Jun JB, Lee HS, Park YW, Im HJ, et al. Analysis of CARD15 polymorphisms in Korean patients with ankylosing spondylitis reveals absence of common variants seen in western populations. J Rheumatol 2004;31(10):1959-61.
. van der Paardt M, Crusius JB, de Koning MH, Murillo LS, van de Stadt RJ, Dijkmans BA, et al. CARD15 gene mutations are not associated with ankylosing spondylitis. Genes Immun 2003;4(1):77-8. http://dx.doi.org/10.1038/sj.gene.6363914.
. Laukens D, Peeters H, Marichal D, Vander Cruyssen B, Mielants H, Elewaut D, et al. CARD15 gene polymorphisms in patients with spondyloarthropathies identify a specific phenotype previously related to Crohn’s disease. Ann Rheum Dis 2005;64(6):930-5. http://dx.doi.org/10.1136/ard.2004.028837.
. Peeters H, Vander Cruyssen B, Laukens D, Coucke P, Marichal D, Van Den Berghe M, et al. Radiological sacroiliitis, a hallmark of spondylitis, is linked with CARD15 gene polymorphisms in patients with Crohn's disease. Ann Rheum Dis 2004;63(9):1131-4. http://dx.doi.org/10.1136/ard.2004.021774.
. Crane AM, Bradbury L, van Heel DA, McGovern DP, Brophy S, Rubin L, et al. Role of NOD2 variants in spondylarthritis. Arthritis Rheum 2002;46(6):1629-33. http://dx.doi.org/10.1002/art.10329.
. Kobak S, Orman M, Berdeli A. NOD2/CARD15 mutations in Turkish patients with ankylosing spondylitis. Scand J Rheumatol 2013;42(3):249-50. http://dx.doi.org/10.3109/03009742.2012.761280.
. Pawlik A, Kurzawski M, Gawronska-Szklarz B, Drozdzik M, Herczynska M. NOD2 allele variants in patients with rheumatoid arthritis. Clin Rheumatol 2007;26(6):868-71. http://dx.doi.org/10.1007/s10067-006-0404-1.
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2016-05-21
Published 2016-11-10









