Immunohistochemical localization of ionotropic glutamate receptors in the rat red nucleus
DOI:
https://doi.org/10.17305/bjbms.2016.1629Keywords:
Red nucleus, parvocellular, magnocellular, rat, glutamate, neuronal function, GluA2, GluA4, GluK2/3, GluK5, GluN1, GluN2A, α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid, kainate, N-methyl-D-aspartate, immunohistochemistryAbstract
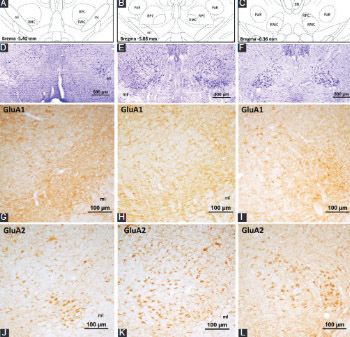
In this study, we aimed to determine the presence as well as the diverse distribution of N-methyl-D-aspartate (NMDA) and non-NMDA glutamate receptor subunits in the rat red nucleus. Using adult Sprague-Dawley rats as the experimental animals, immunohistochemistry was performed on 30 µm thick coronal brain sections with antibodies against α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (GluA1-4), kainate (GluK1, GluK2/3, and GluK5), and NMDA (GluN1 and GluN2A) receptor subunits. The results showed that all ionotropic glutamate receptor subunits are expressed in the red nucleus. Specific staining was localized in the neuron bodies and processes. However, the pattern of immunoreactivity and the number of labeled neurons changed depending on the type of ionotropic glutamate receptor subunits and the localization of neurons in the red nucleus. The neurons localized in the magnocellular part of the red nucleus were particularly immunopositive for GluA2, GluA4, GluK2/3, GluK5, GluN1, and GluN2A receptor proteins. In the parvocellular part of the red nucleus, ionotropic glutamate receptor subunit immunoreactivity of variable intensity (lightly to moderately stained) was detected in the neurons. These results suggest that red nucleus neurons in rat heterogeneously express ionotropic glutamate receptor subunits to form functional receptor channels. In addition, the likelihood of the coexpression of different subunits in the same subgroup of neurons suggests the formation of receptor channels with diverse structure by way of different subunit combination, and the possibility of various neuronal functions through these channels in the red nucleus.
Citations
Downloads
References
Houk JC. Red nucleus: Role in motor control. Curr Opin Neurobiol 1991;1(4):610-5.
Ruigrok TJH. Precerebellar nuclei and red nucleus. In: Paxinos G, editor. The Rat Nervous System. 3rd ed. Amsterdam: Elsevier Academic Press; 2004. p. 167-204. http://dx.doi.org/10.1016/B978-012547638-6/50009-2.
Herter TM, Takei T, Munoz DP, Scott SH. Neurons in red nucleus and primary motor cortex exhibit similar responses to mechanical perturbations applied to the upper-limb during posture. Front Integr Neurosci 2015;9:29. http://dx.doi.org/10.3389/fnint.2015.00029.
Morris R, Vallester KK, Newton SS, Kearsley AP, Whishaw IQ. The differential contributions of the parvocellular and the magnocellular subdivisions of the red nucleus to skilled reaching in the rat. Neuroscience 2015;295:48-57. http://dx.doi.org/10.1016/j.neuroscience.2015.03.027.
Peschanski M, Mantyh PW. Efferent connections of the subfascicular area of the mesodiencephalic junction and its possible involvement in stimulation-produced analgesia. Brain Res 1983;263(2):181-90.
http://dx.doi.org/10.1016/0006-8993(83)90311-6.
Hicks TP, Onodera S. The mammalian red nucleus and its role in motor systems, including the emergence of bipedalism and language. Prog Neurobiol 2012;96(2):165-75. http://dx.doi.org/10.1016/j.pneurobio.2011.12.002.
Reid JM, Gwym DG, Flumerfelt BA. A cytoarchitectonic and Golgi study of the red nucleus in the rat. J Comp Neurol 1975;162(3):337-61. http://dx.doi.org/10.1002/cne.901620306. http://dx.doi.org/10.1002/cne.901620305.
Gruber P, Gould DJ. The red nucleus: Past, present, and future. Neuroanatomy 2010;9:1-3.
Satoh Y, Tsuji K, Tsujimura T, Ishizuka K, Inoue M. Suppression of the swallowing reflex by stimulation of the red nucleus. Brain Res Bull 2015;116:25-33. http://dx.doi.org/10.1016/j.brainresbull.2015.05.007.
Kennedy PR. Corticospinal, rubrospinal and rubro-olivary projections: A unifying hypothesis. Trends Neurosci 1990;13(12):474-9.
http://dx.doi.org/10.1016/0166-2236(90)90079-P.
Hollmann M, Heinemann S. Cloned glutamate receptors. Annu Rev Neurosci 1994;17:31-108. http://dx.doi.org/10.1146/annurev.ne.17.030194.000335.
Wisden W, Seeburg PH. Mammalian ionotropic glutamate receptors. Curr Opin Neurobiol 1993;3(3):291-8. http://dx.doi.org/10.1016/0959-4388(93)90120-N.
Collingridge GL, Olsen RW, Peters J, Spedding M. A nomenclature for ligand-gated ion channels. Neuropharmacology 2009;56(1):2-5. http://dx.doi.org/10.1016/j.neuropharm.2008.06.063.
Bettler B, Mulle C. Review: Neurotransmitter receptors. II. AMPA and kainate receptors. Neuropharmacology 1995;34(2):123-39.
http://dx.doi.org/10.1016/0028-3908(94)00141-E.
Lerma J, Paternain AV, Rodríguez-Moreno A, López-García JC. Molecular physiology of kainate receptors. Physiol Rev 2001;81(3):971-98.
Sucher NJ, Awobuluyi M, Choi YB, Lipton SA. NMDA receptors: From genes to channels. Trends Pharmacol Sci 1996;17(10):348-55. http://dx.doi.org/10.1016/S0165-6147(96)80008-3.
Paoletti P, Neyton J. NMDA receptor subunits: Function and pharmacology. Curr Opin Pharmacol 2007;7(1):39-47. http://dx.doi.org/10.1016/j.coph.2006.08.011.
Seeburg PH. The TINS/TiPS lecture. The molecular biology of mammalian glutamate receptor channels. Trends Neurosci 1993;16(9):359-65. http://dx.doi.org/10.1016/0166-2236(93)90093-2.
Nieoullon A, Vuillon-Cacciuttolo G, Dusticier N, Kerkérian L, André D, Bosler O. Putative neurotransmitters in the red nucleus and their involvement in postlesion adaptive mechanisms. Behav Brain Res 1988;28(1-2):163-74. http://dx.doi.org/10.1016/0166-4328(88)90093-9.
Billard JM, Daniel H, Pumain R. Sensitivity of rubrospinal neurons to excitatory amino acids in the rat red nucleus in vivo. Neurosci Lett 1991;134(1):49-52. http://dx.doi.org/10.1016/0304-3940(91)90506-O.
Jiang MC, Alheid GF, Nunzi MG, Houk JC. Cerebellar input to magnocellular neurons in the red nucleus of the mouse: Synaptic analysis in horizontal brain slices incorporating cerebello-rubral pathways. Neuroscience 2002;110(1):105-21. http://dx.doi.org/10.1016/S0306-4522(01)00544-9.
Davies J, Miller AJ, Sheardown MJ. Amino acid receptor mediated excitatory synaptic transmission in the cat red nucleus. J Physiol 1986;376:13-29. http://dx.doi.org/10.1113/jphysiol.1986.sp016139.
Davies J, Qume M, Harris NC. Pharmacological characterisation of excitatory synaptic transmission in the cat red nucleus in vivo. Brain Res 1994;649(1-2):43-52. http://dx.doi.org/10.1016/0006-8993(94)91047-2.
Yang JC, Fan XL, Song XA, Li Q. The role of different glutamate receptors in the mediation of glutamate-evoked excitation of red nucleus neurons after simulated microgravity in rat. Neurosci Lett 2008;448(3):255-9. http://dx.doi.org/10.1016/j.neulet.2008.10.044.
Satoh Y, Yajima E, Ishizuka K, Nagamine Y, Iwasaki S. Modulation of two types of jaw-opening reflex by stimulation of the red nucleus. Brain Res Bull 2013;97:24-31. http://dx.doi.org/10.1016/j.brainresbull.2013.05.007.
Nieoullon A, Kerkerian L, Dusticier N. High affinity glutamate uptake in the red nucleus and ventrolateral thalamus after lesion of the cerebellum in the adult cat: Biochemical evidence for functional changes in the deafferented structures. Exp Brain Res 1984;55(3):409-19. http://dx.doi.org/10.1007/BF00235271.
Bromberg MB, Pamel G, Stephenson BS, Young AB, Penney JB. Evidence for reactive synaptogenesis in the ventrolateral thalamus and red nucleus of the rat: Changes in high affinity glutamate uptake and numbers of corticofugal fiber terminals. Exp Brain Res 1987;69(1):53-9. http://dx.doi.org/10.1007/BF00247028.
Keifer J, Carr MT. Immunocytochemical localization of glutamate receptor subunits in the brain stem and cerebellum of the turtle Chrysemys picta. J Comp Neurol 2000;427(3):455-68.
http://dx.doi.org/10.1002/1096-9861(20001120)427:3<455::AID-CNE11>3.0.CO;2-X.
Sato K, Kiyama H, Tohyama M. The differential expression patterns of messenger RNAs encoding non-N-methyl-D-aspartate glutamate receptor subunits (GluR1-4) in the rat brain. Neuroscience 1993;52(3):515-39.
http://dx.doi.org/10.1016/0306-4522(93)90403-3.
Eyigor O, Jennes L. Identification of glutamate receptor subtype mRNAs in gonadotropin-releasing hormone neurons in rat brain. Endocrine 1996;4(2):133-9. http://dx.doi.org/10.1007/BF02782758.
Eyigor O, Jennes L. Identification of kainate-preferring glutamate receptor subunit GluR7 mRNA and protein in the rat median eminence. Brain Res 1998;814(1-2):231-5. http://dx.doi.org/10.1016/S0006-8993(98)01056-7.
Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 6th ed. Amsterdam: Elsevier Academic Press; 2006.
Martin LJ, Blackstone CD, Levey AI, Huganir RL, Price DL. AMPA glutamate receptor subunits are differentially distributed in rat brain. Neuroscience 1993;53(2):327-58. http://dx.doi.org/10.1016/0306-4522(93)90199-P.
Petralia RS, Wenthold RJ. Light and electron immunocytochemical localization of AMPA-selective glutamate receptors in the rat brain. J Comp Neurol 1992;318(3):329-54. DOI: 10.1002/cne.903180309.
Petralia RS, Wang YX, Wenthold RJ. The NMDA receptor subunits NR2A and NR2B show histological and ultrastructural localization patterns similar to those of NR1. J Neurosci 1994;14(10):6102-20. http://dx.doi.org/10.1002/cne.903180309.
Petralia RS, Yokotani N, Wenthold RJ. Light and electron microscope distribution of the NMDA receptor subunit NMDAR1 in the rat nervous system using a selective anti-peptide antibody. J Neurosci 1994;14(2):667-96.
Downloads
Additional Files
Published
How to Cite
Accepted 2016-09-28
Published 2017-02-21









