C - reactive protein and chitinase 3-like protein 1 as biomarkers of spatial redistribution of retinal blood vessels on digital retinal photography in patients with diabetic retinopathy
DOI:
https://doi.org/10.17305/bjbms.2014.3.21Keywords:
diabetic retinopathy, YKL-40, CRPAbstract
The aim of the study was to investegate the correlation between the levels of CRP and YKL-40 in blood samples with morphometric parameters of retinal blood vessels in patients with diabetic retinopathy.
Blood laboratory examination of 90 patients included the measurement of glycemia, HbA1C, total cholesterol, LDL-C, HDL-C, triglycerides and CRP. Levels of YKL-40 were detected and measured in serum by ELISA (Micro VueYKL-40 EIA Kit, Quidel Corporation, San Diego, USA).
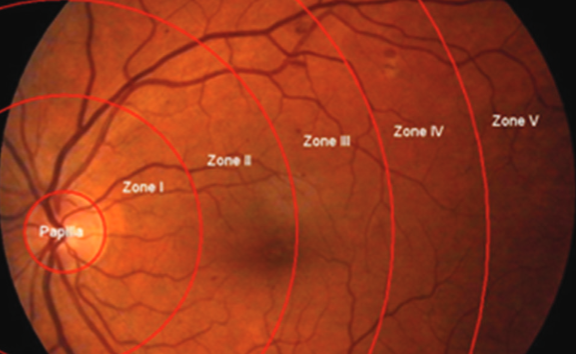
Morphmetric analysis was performed with ImageJ software (http://rsbweb.nih.gov/ij/) for digital retinal photography. We measured the number, diameter of retinal blood vessels in five different parts concentric to the optic disc. Differences between the morphometric parameters and the blood test analysis results were evaluated using the Student’s t – test. One Way ANOVA was used to establish the significance of differences.
CRP and YKL-40 levels were moderately higher in the group of patients with severe diabetic retinopathy. Levels of YKL-40 correlated positively with diameter and negatively with number of retinal blood vessels. The average number of the blood vessels per retinal zone was significantly higher in the group of patients with mild non-proliferative diabetic retinopathy than in the group with severe form in the optic disc and all five retinal zones. The average outer diameter of the evaluated retinal zones and optic disc vessels was significantly higher in the group with severe compared to the group with mild diabetic retinopathy.
Morphological analysis of the retinal vessels on digital fundus photography and correlation with YKL-40 may be valuable for the follow-up of diabetic retinopathy.
Citations
Downloads
References
International Diabetes Federation. IDF Diabetes Atlas, 6th edn. Brussels, Belgium: International Diabetes Federation, 2013.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013; 33:67-74.
Diabetic Retinopathy Guidelines. 2012, The Royal College of Ophthalmologists
Wong TY, Kamineni A, Klein R, Sharrett AR, Klein BE, Siscovick DS, Cushman M, Duncan BB. Quantitative retinal venular caliber and risk of cardio¬vascular disease in older persons. The cardiovascular health study. Arch Intern Med 2006; 166: 2388-94.
Mitchell P, Leung H, Wang JJ, Rochtchina E, Lee A, Wong TY, Klein R. Retinal vessel dia¬meter and open-angle glaucoma: The Blue Mountains Eye Study. Ophthalmology 2005; 112 (2):245-50.
Wong TY, Shankar A, Klein R, Klein BE, Hubbard LD. Retinal arteriolar narrowing, hyper¬ten¬sion, and subse¬qu¬ent risk of diabetes mellitus. Arch Intern Med 2005; 165:1060-5.
Klein R, Klein BE, Moss SE, Wong TY. Retinal vessel caliber and microvascular and macro¬vascular disease in type 2 diabetes: XXI: The Wisconsin epidemiologic study of diabetic reti¬no¬pathy. Ophthalmol 2007; 114; 10:1884-92.
Cheung N, Rogers SL, Donaghue KC, Jenkins AJ,Tikellis G,Wong TY. Retinal arteriolar dilation predicts retinopathy in adolescents with type 1 diabetes. Diabetes Care. 2008; 31(9):1842-6.
Nguyen TT,Wong TY. Retinal vascular changes and diabetic retinopathy. Curr Diab Rep. 2009; 9 (4):277-83.
Nguyen TT, Islam FM, Farouque HM, Klein R, Klein BE, Cotch MF, Herrington DM, Wong TY.Retinal vascular caliber and brachial flow-mediated dilation: the Multi-Ethnic Study of Atherosclerosis. Stroke. 2010; 41(7):1343-8.
Ding J, Ikram MK, Cheung CY, Wong TY. Retinal vascular calibre as predictor of incidence and progression of diabetic retinopathy. Cilin Exp Optom. 2012; 95 (3): 290-296.
Ikram MK, Cheung CY, Lorenzi M, Klein R, Jones TLZ, Wong TY, NIH/JDRF Workshop on Retinal Biomarker for Diabetes Group. Retinal vascular caliber as a biomarker for diabetes microvascular complications. Diabetes Care. 2013; 36(3):750-9.
Joussen AM, Poulaki V, Le ML, Koizumi K, Esser C, Janicki H, Schraermeyer U, Kociok N, Fauser S, Kirchhof B, Kern TS, Adamis AP. A central role of inflammation in the patho¬genesis of diabetic retinopathy. FASEB J 2004; 18(12):1450-2.
Nguyen TT, Alibrahim E, Islam FM, Klein R, Klein BE, Cotch MF, Shea S,Wong TY. Inflammatory, hemostatic, and other novel biomarkers for diabetic retinopathy: the multi-ethnic study of atherosclerosis. Diabetes Care. 2009; 32(9):1704-9.
Rangassamy S, McGuire PG, Das A. Diabetic Retinopathy and Inflammation: Novel Therapeutic Targets. Hum Hypertens. 2010; 24(11):755-762.
Muni RH, Kohly RP, Quant EC, Manson JE, Semba RD, Schaumberg DA. A prospective study of biomarkers and risk of diabetic retinopathy in diabetes control and complications trials. JAMA Ophthalmol. 2013; 131(4):514-521.
Zorena K, Raczyńska D, Raczyńska K. Biomarkers in diabetic retinopathy and the therapeutic implications. Mediators Inflamm. 2013; 2013:193604.
Tomić M, Ljubić S, Kaštelan S, Gverović Antunica A, Jazbec A, Poljičanin T. Inflammation, haemostatic disturbance, and obesity: possible link to pathogenesis of diabetic retinopathy in type 2 diabetes. Mediators Inflamm. 2013; 2013:818671.
Začiragić A, Huskić J, Mulabegović N, Avdagić N, Valjevac A, Hasić S. et al. An assessment of correlation between serum asymmetric dimethylarginine and glycated hemoglobin in patients with type 2 diabetes mellitus. Bosn J Basic Med Sci 2014; 14(4): 21-24.
Kozakova M, Sarafian VS. YKL-40- A Novel Biomarker in Clinical Practice? Folia Med. 2009; 1:5-13.
Johansen J. Study on serum YKL-40 as biomarker in disease with inflammation, tissue remodeling, fibrosis and cancer. Dan Med Bull 2006; 53 172-209.
Streja D, Cressey P, Rabkin SW. Association between inflammatory markers, traditional risk factors, and complications in patients with type 2 diabetes mellitus. J Diab Compl 2003; 17: 120-127.
Hung MJ, Hsu KH, Hu WS, Chang NC, Hung MY. C-reactive protein for predicting prognosis and its gender – specific association with diabetes mellitus and hypertension in development of coronary artery spasm. PLoS One. 2013; 8(10):e77655.
Folsom AR, Wu KK, Rasmunsen M, Chambless LE, Aleksić N, Nieto FJ. Determinants in fibrinogen and factor VII over six yeras: the Atherosclerosis Risk in Communities (ARIC) Study. Arterioscler Throm Vasc Biol 2000; 20:601-606.
Schram MT, Chaturvedi N, Schalkwijk CG, Fuller JH, Stehouwer CD. Markers of infla-mma¬tion are cross-sectionally associated with microvascular complications and cardiovascular disease in type 1 diabetes: EURODIAB Prospective Complications Study. Diabetologia 2005; 48:370-378.
Johansen J, Lotenburger T, Nilsen JH, et al. Diural,Weekly and Long –Time Variation in Serum Concentration of YKL-40 in Healthy Subject. Cancer Epidemiol Biomarkers Prev. 2008; 17(10):2603-2860.
Rathcke CN, Johansen JS, Vestergaard H. YKL-40, biomarker of inflammation, is elevated in patients with type 2 diabetes and is related to insulin resistence. Inflam Res. 2006; 55: 53-59.
Nelsen AR, Erikstrup C, Johansen J, et al. Plasma YKL-40 A BMI – Independent Marker of Type 2 Diabetes. Diab. 2008; 57: 3078-3082.
Sakamoto F, Katakami N, Kaneto H, Yasuda T, Takahara M, Miyashita K, Kuroda A, Matsuhisa M, Kosugi K, Shimomura I. Association of serum YKL-40 levels with urinary albumin excretion rate in young Japanese patients with type 1 diabetes mellitus. Endocr J. 2013; 60:73-9.
Røndbjerg AK, Omerovic E, Vestergaard H.YKL-40 levels are independently associated with albuminuria in type 2 diabetes. Cardiovasc Diabetol. 2011; 10:54-9.
International Diabetes Federation. IDF Diabetes Atlas, 6th edn. Brussels, Belgium: International Diabetes Federation, 2013.
2. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013; 33:67-74
2. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013; 33:67-74.
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2013; 33:67-74.
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2015 Bosnian Journal of Basic Medical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2014-07-19
Published 2014-08-20









