Evaluation of platelet activation in leukocyte-depleted platelet concentrates during storage
DOI:
https://doi.org/10.17305/bjbms.2017.2321Keywords:
Platelet storage, platelet activation markers, leukocyte depletion, in-line filtrationAbstract
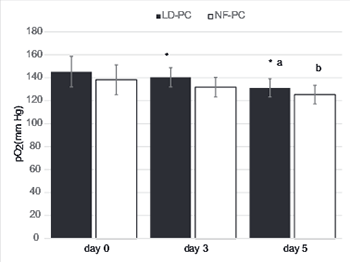
Structural and functional changes in platelets during storage can lead to the loss of platelet reactivity and response. Our aim was to evaluate leukocyte-depleted platelet concentrates on storage days 0, 3 and 5, obtained by in-line filtration. In non-filtered platelet concentrates (NF-PC) group, 180 whole blood units were collected with quadruple blood bags and then compared to another group of 180 whole blood units (leukocyte-depleted platelet concentrates [LD-PC]), collected in Imuflex Whole Blood Filter Saving Platelets (WB-SP) bags with an integrated leukoreduction filter, with regard to the platelet quality and characteristics. The efficacy of the two techniques for platelet concentrate preparation was evaluated by white blood cell (WBC) and platelet count on day 0. The partial pressure of oxygen (pO2), pH, platelets positive for P-selectin (CD62P), CD63, cluster of differentiation 42b (CD42b), phosphatidylserine (PS), and mitochondrial membrane potential (MMP) were analyzed during the storage in both groups. A significantly lower WBC count and higher platelet count was observed in LD-PC compared to NF-PC group, indicating the overall efficacy of the first technique. During the 5-day storage, pH and pO2 decreased in both groups. In LD-PC group, higher pH, increased pO2 and decreased platelet surface expression of CD62P, CD63 and PS were observed compared to NF-PC group. In both groups, the percentage of CD42b positive platelets and MMP did not change significantly during the 5-day period. The assessment of different markers of platelet activation may be an effective tool in evaluating the quality of platelets during storage. A better understanding of platelet activation may provide new insights for developing a novel therapeutic approach in the manipulation of platelet aggregation.
Citations
Downloads
References
Ohto H, Nollet KE. Overview on platelet preservation: Better controls over storage lesion. Transfus Apher Sci 2011;44(3):321-5. https://doi.org/10.1016/j.transci.2011.03.008.
Springer DL, Miller JH, Spinelli SL, Pasa-Tolic L, Purvine SO, Daly DS, et al. Platelet proteome changes associated with diabetes and during platelet storage for transfusion. J Proteome Res 2009;8(5):2261-72. https://doi.org/10.1021/pr800885j.
Heal JM, Phipps RP, Blumberg N. One big unhappy family: Transfusion alloimmunization, thrombosis, and immune modulation/inflammation. Transfusion 2009;49(6):1032-6. https://doi.org/10.1111/j.1537-2995.2009.02182.x.
Slichter SJ, Corson J, Jones MK, Christoffel T, Pellham E, Bailey SL, et al. Exploratory studies of extended storage of apheresis platelets in a platelet additive solution (PAS). Blood 2014;123(2):271-80. https://doi.org/10.1182/blood-2013-05-501247.
Andreu G, Vasse J, Sandid I, Tardivel R, Semana G. Use of random versus apheresis platelet concentrates. Transfus Clin Biol 2007;14(6):514-21. https://doi.org/10.1016/j.tracli.2008.01.004.
Lannan KL, Refaai MA, Ture SK, Morrell CN, Blumberg N, Phipps RP, et al. Resveratrol preserves the function of human platelets stored for transfusion. Br J Haematol 2016;172(5):794-806. https://doi.org/10.1111/bjh.13862.
Ferrer F, Rivera J, Corral J, González-Conejero R, Vicente V. Evaluation of leukocyte-depleted platelet concentrates obtained by in-line filtration. Vox Sang 2000;78(4):235-41. https://doi.org/10.1046/j.1423-0410.2000.7840235.x.
Apelseth TO, Hervig T. In vitro evaluation of platelet concentrates during storage: Platelet counts and markers of platelet destruction. Transfus Apher Sci 2007;37(3):261-8. https://doi.org/10.1016/j.transci.2007.02.006.
Tynngard N, Wallstedt M, Södergren AL, Faxälv L, Ramström S. Platelet adhesion changes during storage studied with a novel method using flow cytometry and protein-coated beads. Platelets 2015;26(2):177-85. https://doi.org/10.3109/09537104.2014.891728.
Paunovic D, van der Meer P, Kjeldsen-Kragh J, Kekomaki R, Larsson S, Greppi N, et al. Multicenter evaluation of a whole-blood filter that saves platelets. Transfusion 2004;44(8):1197-203. https://doi.org/10.1111/j.1537-2995.2004.03350.x.
Mokhtar MB, Hashim HB, Joshi SR. Assessment of quality of platelets preserved in plasma and platelet additive solution: A Malaysian experience. Asian J Transfus Sci 2016;10(1):84-7. https://doi.org/10.4103/0973-6247.172177.
Skripchenko A, Gelderman MP, Awatefe H, Turqeon A, Thompson-Montgomery D, Cheng C, et al. Automated cold temperature cycling improves platelet properties and in vivo recovery in a mouse model compared to continuous cold storage. Transfusion 2016;56(1):24-32. https://doi.org/10.1111/trf.13273.
Hosseini E, Ghasemzadeh M, Nassaji F, Jamaat ZP. GPVI modulation during platelet activation and storage: Its expression levels and ectodomain shedding compared to markers of platelet storage lesion. Platelets 2017;28(5):498-508. https://doi.org/10.1080/09537104.2016.1235692.
Pavlovic V, Cekic S, Ciric M, Krtinic D, Jovanovic J. Curcumin attenuates Mancozeb-induced toxicity in rat thymocytes through mitochondrial pathway. Food Chem Toxicol 2016;88:105-11. https://doi.org/10.1016/j.fct.2015.12.029.
Heddle NM, Barty RL, Sigouin CS, Boye DM, Nelson EJ, Blajchman MA, et al. In vitro evaluation of prestorage pooled leukoreduced whole bloodderived platelets stored for up to 7 days. Transfusion 2005;45(6):904-10. https://doi.org/10.1111/j.1537-2995.2005.04234.x.
Glas M, Bauer JV, Eichler H, Volk T. Impedance aggregometric analysis of platelet function of apheresis platelet concentrates as a function of storage time. Scand J Clin Lab Invest 2016;76(8):664-70. https://doi.org/10.1080/00365513.2016.1238505.
Zhang JG, Carter CJ, Culibrk B, Devine DV, Levin E, Scammell K, et al. Buffy-coat platelet variables and metabolism during storage in additive solutions or plasma. Transfusion 2008;48(5):847-56. https://doi.org/10.1111/j.1537-2995.2008.01645.x.
Slichter SJ, Bolgiano D, Jones MK, Christoffel T, Corson J, Rose L, et al. Viability and function of 8-day-stored apheresis platelets. Transfusion 2006;46(10):1763-9. https://doi.org/10.1111/j.1537-2995.2006.00970.x.
Johnson L, Schubert P, Tan S, Devine DV, Marks DC. Extended storage and glucose exhaustion are associated with apoptotic changes in platelets stored in additive solution. Transfusion 2016;56(2):360-8. https://doi.org/10.1111/trf.13345.
Tudisco C, Jett BW, Byrne K, Oblitas J, Leitman SF, Stroncek DF. The value of pH as a quality control indicator for apheresis platelets. Transfusion 2005;45(5):773-8. https://doi.org/10.1111/j.1537-2995.2005.04344.x.
Ekaney ML, Grable MA, Powers WF 4th, McKillop IH, Evans SL. Cytochrome c and resveratrol preserve platelet function during cold storage. J Trauma Acute Care Surg 2017;83(2): 271-7. https://doi.org/10.1097/TA.0000000000001547.
Beard MJ, Jeewa Z, Bashir S, Cardigan R, Thomas S. Comparison of platelet activation in platelet concentrates measured by flow cytometry or ADVIA 2120. Vox Sang 2011;101(2):122-30. https://doi.org/10.1111/j.1423-0410.2011.01469.x.
Plaza EM, Lozano ML, Guiu IS, Egea JM, Vicente V, de Terán LC, et al. Evaluation of platelet function during extended storage in additive solution, prepared in a new container that allows manual buffy-coat platelet pooling and leucoreduction in the same system. Blood Transfus 2012;10(4):480-9. DOI: 10.2450/2012.0112-11.
van Velzen JF, Laros-van Gorkom BA, Pop GA, van Heerde WL. Multicolor flow cytometry for evaluation of platelet surface antigens and activation markers. Thromb Res 2012;130(1):92-8. https://doi.org/10.1016/j.thromres.2012.02.041.
Ahmed AS, Leheta O, Younes S. In vitro assessment of platelet storage lesions in leucoreduced random donor platelet concentrates. Blood Transfus 2010;8(1):28-35. DOI: 10.2450/2009.0077-09.
Morrison A, McMillan L, Radwanski K, Blatchford O, Min K, Petrik J. Storage of apheresis platelet concentrates after manual replacement of >95% of plasma with PAS 5. Vox Sang 2014;107(3):247-53. https://doi.org/10.1111/vox.12157.
Albanyan AM, Harrison P, Murphy MF. Markers of platelet activation and apoptosis during storage of apheresis- and buffy coat-derived platelet concentrates for 7 days. Transfusion 2009;49(1):108-17. https://doi.org/10.1111/j.1537-2995.2008.01942.x.
Sandgren P, Hansson M, Gulliksson H, Shanwell A. Storage of buffy-coat-derived platelets in additive solutions at 4 degrees C and 22 degrees C: Flow cytometry analysis of platelet glycoprotein expression. Vox Sang 2007;93(1):27-36.
https://doi.org/10.1111/j.1423-0410.2007.00912.x.
Cookson P, Sutherland J, Turner C, Bashir S, Wiltshire M, Hancoc V, et al. Platelet apoptosis and activation in platelet concentrates stored for up to 12 days in plasma or additive solution. Transfus Med 2010;20(6):392-402. https://doi.org/10.1111/j.1365-3148.2010.01034.x.
Albanyan AM, Murphy MF, Rasmussen JT, Heegaard CW, Harrison P. Measurement of phosphatidylserine exposure during storage of platelet concentrates using the novel probe lactadherin: A comparison study with annexin V. Transfusion 2009;49(1):99-107. https://doi.org/10.1111/j.1537-2995.2008.01933.x.
Saunders C, Rowe G, Wilkins K, Holme S, Collins P. In vitro storage characteristics of platelet concentrates suspended in 70% SSP+(TM) additive solution versus plasma over a 14-day storage period. Vox Sang 2011;101(2):112-21.
https://doi.org/10.1111/j.1423-0410.2011.01468.x.
Gyulkhandanyan AV, Mutlu A, Freedman J, Leytin V. Markers of platelet apoptosis: Methodology and applications. J Thromb Thrombolysis 2012;33(4):397-411. https://doi.org/10.1007/s11239-012-0688-8.
Perrotta PL, Perrotta CL, Snyder EL. Apoptotic activity in stored human platelets. Transfusion 2003;43(4):526-35. https://doi.org/10.1046/j.1537-2995.2003.00349.x.
Leytin V, Allen DJ, Mutlu A, Mykhaylov S, Lyubimov E, Freedman J. Platelet activation and apoptosis are different phenomena: Evidence from the sequential dynamics and the magnitude of responses during platelet storage. Br J Haematol 2008;142(3):494-7. https://doi.org/10.1111/j.1365-2141.2008.07209.x.
Downloads
Additional Files
Published
How to Cite
Accepted 2017-08-30
Published 2018-02-20









