MicroRNA-466 (miR-466) functions as a tumor suppressor and prognostic factor in colorectal cancer (CRC)
DOI:
https://doi.org/10.17305/bjbms.2018.2376Keywords:
miR-466, colorectal cancer, CRC, prognosis, proliferation, migration, invasionAbstract
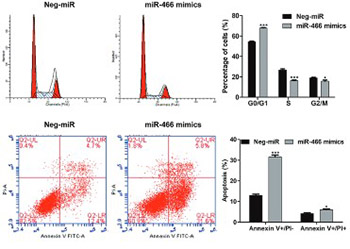
MicroRNAs (miRNAs) have an important role in the regulation of tumor development and metastasis. In this study, we investigated the clinical and prognostic value as well as biological function of miR-466 in colorectal cancer (CRC). Tumor and adjacent healthy tissues were obtained from 100 patients diagnosed with CRC. miR-466 expression was determined by quantitative reverse transcription polymerase chain reaction (qRT-PCR). mRNA and protein levels of cyclin D1, apoptosis regulator BAX (BAX), and matrix metalloproteinase-2 (MMP-2) were analyzed by qRT-PCR and Western blot, respectively, in SW-620 CRC cells transfected with miR-466 mimics or negative control miRNA. Effects of miR-466 on SW-620 cell proliferation, cell cycle and apoptosis, and invasion were investigated using CCK-8 assay, flow cytometry and Transwell assay, respectively. miR-466 expression was significantly downregulated in tumor tissues compared to matched adjacent non-tumor tissues. Low expression of miR-466 was significantly correlated with the tumor size, Tumor Node Metastasis stage, lymph node metastasis, and distant metastasis. The overall survival of CRC patients with low miR-466 expression was significantly shorter compared to high-miR-466 expression group (log-rank test: p = 0.0103). Multivariate analysis revealed that low miR-466 expression was associated with poor prognosis in CRC patients. The ectopic expression of miR-466 suppressed cell proliferation and migration/invasion, as well as induced G0/G1 arrest and apoptosis in SW-620 cells. Moreover, the ectopic expression of miR-466 decreased the expression of cyclin D1 and MMP-2, but increased BAX expression in SW-620 cells. In conclusion, our findings demonstrated that miR-466 functions as a suppressor miRNA in CRC and may be used as a prognostic factor in these patients.
Citations
Downloads
References
Giantonio BJ, Catalano PJ, Meropol NJ, O'Dwyer PJ, Mitchell EP, Alberts SR, et al. Bevacizumab in combination with oxaliplatin, fluorouracil, and leucovorin (FOLFOX4) for previously treated metastatic colorectal cancer: Results from the Eastern Cooperative Oncology Group Study E3200. J Clin Oncol 2007 20;25(12):1539-44. https://doi.org/10.1200/JCO.2006.09.6305.
Siegel R, Desantis C, Jemal A. Colorectal cancer statistics, 2014. CA Cancer J Clin 2014;64(2):104-17. https://doi.org/10.3322/caac.21220.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin 2016;66(1):7-30. https://doi.org/10.3322/caac.21332.
Haggar FA, Boushey RP. Colorectal cancer epidemiology: Incidence, mortality, survival, and risk factors. Clin Colon Rectal Surg 2009;22(4):191-7.
https://doi.org/10.1055/s-0029-1242458.
Schaefer A, Jung M, Mollenkopf HJ, Wagner I, Stephan C, Jentzmik F, et al. Diagnostic and prognostic implications of microRNA profiling in prostate carcinoma. Int J Cancer 2010;126(5):1166-76. DOI: 10.1002/ijc.24827.
Filipowicz W, Bhattacharyya SN, Sonenberg N. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight? Nat Rev Genet 2008;9(2):102-14. https://doi.org/10.1038/nrg2290.
Volinia S, Calin GA, Liu CG, Ambs S, Cimmino A, Petrocca F, et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proc Natl Acad Sci U S A 2006;103(7):2257-61. https://doi.org/10.1073/pnas.0510565103.
Lin M, Duan B, Hu J, Yu H, Sheng H, Gao H, et al. Decreased expression of miR-193a-3p is associated with poor prognosis in colorectal cancer. Oncol Lett 2017;14(1):1061-7. https://doi.org/10.3892/ol.2017.6266.
Cao J, Yan XR, Liu T, Han XB, Yu JJ, Liu SH, et al. MicroRNA-552 promotes tumor cell proliferation and migration by directly targeting DACH1 via the Wnt/beta-catenin signaling pathway in colorectal cancer. Oncol Lett 2017;14(3):3795-802. DOI: 10.3892/ol.2017.6600.
Jiang H, Liu J, Chen Y, Ma C, Li B, Hao T. Up-regulation of mir-10b predicate advanced clinicopathological features and liver metastasis in colorectal cancer. Cancer Med 2016;5(10):2932-41. https://doi.org/10.1002/cam4.789.
Colden M, Dar AA, Saini S, Dahiya PV, Shahryari V, Yamamura S, et al. MicroRNA-466 inhibits tumor growth and bone metastasis in prostate cancer by direct regulation of osteogenic transcription factor RUNX2. Cell Death Dis 2017;8(1):e2572. https://doi.org/10.1038/cddis.2017.15.
Sun P, Shen Y, Gong JM, Zhou LL, Sheng JH, Duan FJ. A new microRNA expression signature for cervical cancer. Int J Gynecol Cancer 2017;27(2):339-43. https://doi.org/10.1097/IGC.0000000000000863.
Martini G, Troiani T, Cardone C, Vitiello P, Sforza V, Ciardiello D, et al. Present and future of metastatic colorectal cancer treatment: A review of new candidate targets. World J Gastroenterol 2017;23(26):4675-88. https://doi.org/10.3748/wjg.v23.i26.4675.
Atreya CE, Yaeger R, Chu E. Systemic therapy for metastatic colorectal cancer: From current standards to future molecular targeted approaches. Am Soc Clin Oncol Educ Book 2017;37:246-56. https://doi.org/10.14694/EDBK_175679.
Nilsson I, Hoffmann I. Cell cycle regulation by the Cdc25 phosphatase family. Prog Cell Cycle Res 2000;4:107-14. https://doi.org/10.1007/978-1-4615-4253-7_10.
Gyuris J, Golemis E, Chertkov H, Brent R. Cdi1, a human G1 and S phase protein phosphatase that associates with Cdk2. Cell 1993;75(4):791-803. https://doi.org/10.1016/0092-8674(93)90498-F.
Yin XM. Signal transduction mediated by Bid, a pro-death Bcl-2 family proteins, connects the death receptor and mitochondria apoptosis pathways. Cell Res 2000;10(3):161-7. https://doi.org/10.1038/sj.cr.7290045.
Jürgensmeier JM, Xie Z, Deveraux Q, Ellerby L, Bredesen D, Reed JC. Bax directly induces release of cytochrome c from isolated mitochondria. Proc Natl Acad Sci U S A 1998;95(9):4997-5002. https://doi.org/10.1073/pnas.95.9.4997.
Raisova M, Hossini AM, Eberle J, Riebeling C, Wieder T, Sturm I, et al. The Bax/Bcl-2 ratio determines the susceptibility of human melanoma cells to CD95/Fas-mediated apoptosis. J Invest Dermatol 2001;117(2):333-40.
https://doi.org/10.1046/j.0022-202x.2001.01409.x
LeBlanc H, Lawrence D, Varfolomeev E, Totpal K, Morlan J, Schow P, et al. Tumor-cell resistance to death receptor - induced apoptosis through mutational inactivation of the proapoptotic Bcl-2 homolog Bax. Nat Med 2002;8(3):274-81. https://doi.org/10.1038/nm0302-274.
Sahai E. Mechanisms of cancer cell invasion. Curr Opin Genet Dev 2005;15(1):87-96. https://doi.org/10.1016/j.gde.2004.12.002.
Wolf K, Wu YI, Liu Y, Geiger J, Tam E, Overall C, et al. Multi-step pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol 2007;9(8):893-904. https://doi.org/10.1038/ncb1616.
Zheng H, Takahashi H, Murai Y, Cui Z, Nomoto K, Niwa H, et al. Expressions of MMP-2, MMP-9 and VEGF are closely linked to growth, invasion, metastasis and angiogenesis of gastric carcinoma. Anticancer Res 2006;26(5A):3579-83.
Oku N, Sasabe E, Ueta E, Yamamoto T, Osaki T. Tight junction protein claudin-1 enhances the invasive activity of oral squamous cell carcinoma cells by promoting cleavage of laminin-5 gamma2 chain via matrix metalloproteinase (MMP)-2 and membrane-type MMP-1. Cancer Res 2006;66(10):5251-7. https://doi.org/10.1158/0008-5472.CAN-05-4478.
Agarwal R, D'Souza T, Morin PJ. Claudin-3 and claudin-4 expression in ovarian epithelial cells enhances invasion and is associated with increased matrix metalloproteinase-2 activity. Cancer Res 2005;65(16):7378-85. https://doi.org/10.1158/0008-5472.CAN-05-1036.
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-09-25
Published 2018-08-01









