Prognostic role of neutrophil-to-lymphocyte ratio (NLR) in patients with operable ampullary carcinoma
DOI:
https://doi.org/10.17305/bjbms.2017.2530Keywords:
Carcinoma of the ampulla of Vater, neutrophil-to-lymphocyte ratio, prognosis, survival, inflammation, NLR, ampullary carcinomaAbstract
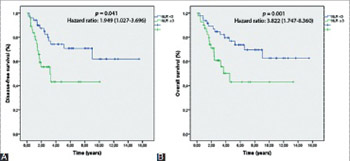
Ampullary carcinoma or cancer of the ampulla of Vater is a rare malignancy with a high recurrence rate. Although cost-effective biomarkers, such as neutrophil-to-lymphocyte ratio (NLR), have been investigated in other cancers for predicting postoperative prognosis in patients, studies on the role of NLR in ampullary cancer are scarce. Here we aimed to evaluate the prognostic significance of preoperative NLR in patients with operable ampullary carcinoma. We retrospectively reviewed 87 patients who underwent pancreaticoduodenectomy for the treatment of ampullary carcinoma between December 1999 and April 2014. The association between NLR and prognosis (overall survival [OS] and disease-free survival [DFS]) was evaluated. Possible correlations between NLR and clinicopathological features were also assessed. The 5-year DFS and OS rates after surgery in patients with ampullary carcinoma were 51% and 63%, respectively. A high NLR (≥3.0) was found in 40 patients. The NLR was a significant prognostic factor for both OS and DFS. Multivariate analysis revealed a significantly worse OS in patients with positive surgical margins and NLR ≥3 (p = 0.001). Patients with T3-T4 stage (p = 0.029) and NLR ≥3 (p = 0.043) had a lower DFS. Patients with a high NLR had a significantly worse Eastern Cooperative Oncology Group performance score. Preoperative NLR is an independent and significant predictive factor of prognosis in patients with ampullary carcinoma. An elevated pretreatment NLR (e.g., NLR ≥3) may be considered as a biomarker for poor prognosis in patients with ampullary carcinoma.
Citations
Downloads
References
Rosenberg R, Friederichs J, Schuster T, Gertler R, Maak M, Becker K, et al. Prognosis of patients with colorectal cancer is associated with lymph node ratio: A single-center analysis of 3,026 patients over a 25-year time period. Ann Surg 2008;248(6):968-78. https://doi.org/10.1097/SLA.0b013e318190eddc.
Yamaguchi K, Enjoji M. Carcinoma of the ampulla of vater. A clinicopathologic study and pathologic staging of 109 cases of carcinoma and 5 cases of adenoma. Cancer 1987;59(3):506-15. https://doi.org/10.1002/1097-0142(19870201)59:3<506::AID-CNCR2820590326>3.0.CO;2-#.
Bhatia S, Miller RC, Haddock MG, Donohue JH, Krishnan S. Adjuvant therapy for ampullary carcinomas: The Mayo clinic experience. Int J Radiat Oncol Biol Phys 2006;66(2):514-9. https://doi.org/10.1016/j.ijrobp.2006.04.018.
Willett CG, Warshaw AL, Convery K, Compton CC. Patterns of failure after pancreaticoduodenectomy for ampullary carcinoma. Surg Gynecol Obstet 1993;176(1):33-8.
Bettschart V, Rahman MQ, Engelken FJ, Madhavan KK, Parks RW, Garden OJ. Presentation, treatment and outcome in patients with ampullary tumours. Br J Surg 2004;91(12):1600-7. https://doi.org/10.1002/bjs.4787.
Colussi O, Voron T, Pozet A, Hammel P, Sauvanet A, Bachet JB, et al. Prognostic score for recurrence after whipple’s pancreaticoduodenectomy for ampullary carcinomas; Results of an AGEO retrospective multicenter cohort. Eur J Surg Oncol 2015;41(4):520-6. https://doi.org/10.1016/j.ejso.2015.01.010.
Miyakawa S, Ishihara S, Horiguchi A, Takada T, Miyazaki M, Nagakawa T, et al. Biliary tract cancer treatment: 5,584 results from the biliary tract cancer statistics registry from 1998 to 2004 in Japan. J Hepatobiliary Pancreat Surg 2009;16(1):1-7. https://doi.org/10.1007/s00534-008-0015-0.
Morris-Stiff G, Alabraba E, Tan YM, Shapey I, Bhati C, Tanniere P, et al. Assessment of survival advantage in ampullary carcinoma in relation to tumour biology and morphology. Eur J Surg Oncol 2009;35(7):746-50. https://doi.org/10.1016/j.ejso.2008.10.010.
Beger HG, Treitschke F, Gansauge F, Harada N, Hiki N, Mattfeldt T, et al. Tumor of the ampulla of vater: Experience with local or radical resection in 171 consecutively treated patients. Arch Surg 1999;134(5):526-32. https://doi.org/10.1001/archsurg.134.5.526.
Sellner FJ, Riegler FM, Machacek E. Implications of histological grade of tumour for the prognosis of radically resected periampullary adenocarcinoma. Eur J Surg 1999;165(9):865-70. https://doi.org/10.1080/11024159950189375.
Bhatti I, Peacock O, Lloyd G, Larvin M, Hall RI. Preoperative hematologic markers as independent predictors of prognosis in resected pancreatic ductal adenocarcinoma: Neutrophil-lymphocyte versus platelet-lymphocyte ratio. Am J Surg 2010;200(2):197-203. https://doi.org/10.1016/j.amjsurg.2009.08.041.
Malietzis G, Giacometti M, Askari A, Nachiappan S, Kennedy RH, Faiz OD, et al. A preoperative neutrophil to lymphocyte ratio of 3 predicts disease-free survival after curative elective colorectal cancer surgery. Ann Surg 2014;260(2):287-92. https://doi.org/10.1097/SLA.0000000000000216.
Mano Y, Shirabe K, Yamashita Y, Harimoto N, Tsujita E, Takeishi K, et al. Preoperative neutrophil-to-lymphocyte ratio is a predictor of survival after hepatectomy for hepatocellular carcinoma: A retrospective analysis. Ann Surg 2013;258(2):301-5. https://doi.org/10.1097/SLA.0b013e318297ad6b.
McMillan DC. An inflammation-based prognostic score and its role in the nutrition-based management of patients with cancer. Proc Nutr Soc 2008;67(3):257-62. https://doi.org/10.1017/S0029665108007131.
Paramanathan A, Saxena A, Morris DL. A systematic review and meta-analysis on the impact of pre-operative neutrophil lymphocyte ratio on long term outcomes after curative intent resection of solid tumours. Surg Oncol 2014;23(1):31-9. https://doi.org/10.1016/j.suronc.2013.12.001.
Haruki K, Shiba H, Horiuchi T, Shirai Y, Iwase R, Fujiwara Y, et al. Neutrophil to lymphocyte ratio predicts therapeutic outcome after pancreaticoduodenectomy for carcinoma of the ampulla of Vater. Anticancer Res 2016;36(1):403-8.
Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell 2010;140(1):883-99. https://doi.org/10.1016/j.cell.2010.01.025.
Halazun KJ, Aldoori A, Malik HZ, Al-Mukhtar A, Prasad KR, Toogood GJ, et al. Elevated preoperative neutrophil to lymphocyte ratio predicts survival following hepatic resection for colorectal liver metastases. Eur J Surg Oncol 2008;34(1):55-60. https://doi.org/10.1016/j.ejso.2007.02.014.
Han S, Liu Y, Li Q, Li Z, Hou H, Wu A, et al. Pre-treatment neutrophil-to-lymphocyte ratio is associated with neutrophil and T-cell infiltration and predicts clinical outcome in patients with glioblastoma. BMC Cancer 2015;15:617.
https://doi.org/10.1186/s12885-015-1629-7.
Na N, Yao J, Cheng C, Huang Z, Hong L, Li H, et al. Meta-analysis of the efficacy of the pretreatment neutrophil-to-lymphocyte ratio as a predictor of prognosis in renal carcinoma patients receiving tyrosine kinase inhibitors. Oncotarget 2016;7(28):44039-46. https://doi.org/10.18632/oncotarget.9836.
Viers BR, Boorjian SA, Frank I, Tarrell RF, Thapa P, Karnes RJ, et al. Pretreatment neutrophil-to-lymphocyte ratio is associated with advanced pathologic tumor stage and increased cancer-specific mortality among patients with urothelial carcinoma of the bladder undergoing radical cystectomy. Eur Urol 2014;66(6):1157-64. https://doi.org/10.1016/j.eururo.2014.02.042.
Yao M, Liu Y, Jin H, Liu X, Lv K, Wei H, et al. Prognostic value of preoperative inflammatory markers in Chinese patients with breast cancer. Onco Targets Ther 2014;7:1743-52. https://doi.org/10.2147/OTT.S69657.
Zhao QT, Yang Y, Xu S, Zhang XP, Wang HE, Zhang H, et al. Prognostic role of neutrophil to lymphocyte ratio in lung cancers: A meta-analysis including 7,054 patients. OncoTargets Ther 2015;8:2731-8. https://doi.org/10.2147/OTT.S90875.
Chen JW, Bhandari M, Astill DS, Wilson TG, Kow L, Brooke-Smith M, et al. Predicting patient survival after pancreaticoduodenectomy for malignancy: Histopathological criteria based on perineural infiltration and lymphovascular invasion. HPB (Oxford) 2010;12(2):101-8. https://doi.org/10.1111/j.1477-2574.2009.00140.x.
Klein F, Jacob D, Bahra M, Pelzer U, Puhl G, Krannich A, et al. Prognostic factors for long-term survival in patients with ampullary carcinoma: The results of a 15-year observation period after pancreaticoduodenectomy. HPB Surg 2014;2014:970234. https://doi.org/10.1155/2014/970234.
Lowe MC, Coban I, Adsay NV, Sarmiento JM, Chu CK, Staley CA, et al. Important prognostic factors in adenocarcinoma of the ampulla of Vater. Am Surg 2009;75:754-60.
Pomianowska E, Westgaard A, Mathisen Ø, Clausen OP, Gladhaug IP. Prognostic relevance of number and ratio of metastatic lymph nodes in resected pancreatic, ampullary, and distal bile duct carcinomas. Ann Surg Oncol 2013;20(1):233-41. https://doi.org/10.1245/s10434-012-2592-z.
Qiao QL, Zhao YG, Ye ML, Yang YM, Zhao JX, Huang YT, et al. Carcinoma of the ampulla of Vater: Factors influencing long-term survival of 127 patients with resection. World J Surg 2007;31(1):137-43. https://doi.org/10.1007/s00268-006-0213-3.
Jass JR. Lymphocytic infiltration and survival in rectal cancer. J Clin Pathol 1986;39(6):585-9. https://doi.org/10.1136/jcp.39.6.585.
Kusumanto YH, Dam WA, Hospers GA, Meijer C, Mulder NH. Platelets and granulocytes, in particular the neutrophils, form important compartments for circulating vascular endothelial growth factor. Angiogenesis 2003;6(4):283-7. https://doi.org/10.1023/B:AGEN.0000029415.62384.ba.
Azab B, Bhatt VR, Phookan J, Murukutla S, Kohn N, Terjanian T, et al. Usefulness of the neutrophil-to-lymphocyte ratio in predicting short- and long-term mortality in breast cancer patients. Ann Surg Oncol 2012;19(1):217-24.
https://doi.org/10.1245/s10434-011-1814-0.
Maniecki MB, Etzerodt A, Ulhoi BP, Steiniche T, Borre M, Dyrskjot L, et al. Tumor-promoting macrophages induce the expression of the macrophage-specific receptor CD163 in malignant cells. Int J Cancer 2012;131(10):2320-31. https://doi.org/10.1002/ijc.27506.
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-11-01
Published 2018-08-01









