Epidermal growth factor (EGF) promotes bone healing in surgically induced osteonecrosis of the femoral head (ONFH)
DOI:
https://doi.org/10.17305/bjbms.2018.3259Keywords:
Osteonecrosis, epidermal growth factor, EGF, osteopontin, OPN, angiogenic agents, core decompression, CD31, cluster of differentiation 31Abstract
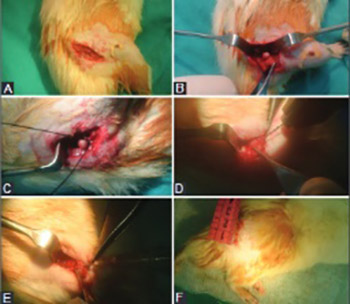
Angiogenic effects of epidermal growth factor (EGF), a potent mitogen, have been demonstrated previously. Moreover, different in vitro studies showed that EGF affects processes associated with bone healing, such as osteoblast differentiation and bone resorption. The aim of this study was to investigate the effect of combined core decompression (CD) and recombinant human EGF (rhEGF) treatment on early-stage osteonecrosis of the femoral head (ONFH) surgically induced in rats. ONFH was induced by dissecting the cervical periosteum and placing a ligature tightly around the femoral neck. Thirty rats were assigned to one of the following groups (n = 10 each group): sham-operated control, CD, and CD+rhEGF group. rhEGF was injected intraosseously into infarcted areas 2 weeks after the surgery. Preservation of femoral head architecture was assessed at 8 weeks post treatment by radiographic and histomorphological analyses. Osteopontin (OPN) and cluster of differentiation 31 (CD31) were detected by immunochemistry, as indicators of bone remodeling and vascular density, respectively. Inter- and intra-group (non-operated left and operated right femur) differences in radiographic and histomorphological results were analyzed. The femoral head area and sphericity were more preserved in CD+rhEGF compared to CD and sham-control group. CD31 levels were significantly different between the three groups, and were higher in CD+rhEGF compared to CD group. OPN levels were increased in CD and CD+rhEGF groups compared to sham control, but with no significant difference between CD and CD+rhEGF groups. Overall, our results indicate that EGF promotes bone formation and microvascularization in ONFH and thus positively affects the preservation of femoral head during healing.
Citations
Downloads
References
Tripathy SK, Goyal T, Sen RK. Management of femoral head osteonecrosis: Current concepts. Indian J Orthop 2015; 49(1):28-45. https://doi.org/10.4103/0019-5413.143911.
Chim SM, Tickner J, Chow ST, Kuek V, Guo B, Zhang G, et al. Angiogenic factors in bone local environment. Cytokine Growth Factor Rev 2013;24(3):297-310. https://doi.org/10.1016/j.cytogfr.2013.03.008.
Matsuda N, Kimar NM, Ramakrishnan PR, Cho M-I. Role of epidermal growth factor receptor in osteoblastic differentiation of rat bone marrow stromal cells. J Bone Miner Metab 1996;14(3):137-45. https://doi.org/10.1007/BF02239481.
Zhang X, Tamasi J, Lu X, Zhu J, Chen H, Tian X, et al. Epidermal growth factor receptor plays an anabolic role in bone metabolism in vivo. J Bone Miner Res 2011;26(5):1022-34. https://doi.org/10.1002/jbmr.295.
Antosz ME, Bellows CG, Aubin JE. Biphasic effects of epidermal growth factor on bone nodule formation by isolated rat calvaria cells in vitro. J Bone Miner Res 1987;2(5):385-93. https://doi.org/10.1002/jbmr.5650020505.
Raisz LG, Simmons HA, Sandberg AL, Canalis E. Direct stimulation of bone resorption by epidermal growth factor. Endocrinology 1980;107(1):270-3.
https://doi.org/10.1210/endo-107-1-270.
Teramatsu Y, Maeda H, Sugii H, Tomokiyo A, Hamano S, Wada N, et al. Expression and effects of epidermal growth factor on human periodontal ligament cells. Cell Tissue Res 2014;357(3):633-43. https://doi.org/10.1007/s00441-014-1877-x.
Park BH, Jang KY, Kim KH, Song KH, Lee SY, Yoon SJ, et al. COMP-Angiopoietin-1 ameliorates surgery-induced ischemic necrosis of the femoral head in rats. Bone 2009;44(5):886-92. https://doi.org/10.1016/j.bone.2009.01.366.
Konturek S, Dembinski A, Warzecha Z, Brzozowski T, Gregory H. Role of epidermal growth factor in healing of chronic gastroduodenal ulcers in rats. Gastroenterology 1988;94(6):1300-7. https://doi.org/10.1016/0016-5085(88)90667-1.
Sarıkaya B, Yumuşak N, Yigin A, Sipahioğlu S, Yavuz Ü, Altay MA. Comparison of the effects of human recombinant epidermal growth factor and platelet-rich plasma on healing of rabbit patellar tendon. Eklem Hastalik Cerrahisi 2017;28(2):92-9. https://doi.org/10.5606/ehc.2017.55396.
Mose K. Methods of measuring in Legg-Calve-Perthes disease with special regard to the prognosis. Clin Orthop Relat Res 1980;(150):103-9.
Hopson CN, Siverhus SW. Ischemic necrosis of the femoral head. Treatment by core decompression. J Bone Joint Surg Am 1988;70(7):1048-51.
Lieberman JR, Berry DJ, Mont MA, Aaron RK, Callaghan JJ, Rajadhyaksha AD, et al. Osteonecrosis of the hip: Management in the 21st century. Instr Course Lect 2003;52:337-55.
Classen T, Becker A, Landgraeber S, Haversath M, Li X, Zilkens C, et al. Long-term clinical results after iloprost treatment for bone marrow edema and avascular necrosis. Orthop Rev (Pavia) 2016;8(1):6150. https://doi.org/10.4081/or.2016.6150.
Nozaki Y, Kumagai K, Miyata N, Niwa M. Pravastatin reduces steroid-induced osteonecrosis of the femoral head in SHRSP rats. Acta Orthop 2012;83(1):87-92. https://doi.org/10.3109/17453674.2011.641103.
Peled E, Bejar J, Zinman C, Reis DN, Boss JH, Ben-Noon H, et al. Alendronate preserves femoral head shape and height/length ratios in an experimental rat model: A computer-assisted analysis. Indian J Orthop 2009;43(1):22-6. https://doi.org/10.4103/0019-5413.44630.
Zhao DW, Yu XB. Core decompression treatment of early-stage osteonecrosis of femoral head resulted from venous stasis or artery blood supply insufficiency. J Surg Res 2015;194(2):614-21. https://doi.org/10.1016/j.jss.2014.12.007.
Norman D, Miller Y, Sabo E, Misselevich I, Peskin B, Zinman C, et al. The effects of enoxaparin on the reparative processes in experimental osteonecrosis of the femoral head of the rat. APMIS 2002;110(3):221-8. https://doi.org/10.1034/j.1600-0463.2002.100304.x.
Ding S, Peng H, Fang HS, Zhou JL, Wang Z. Pulsed electromagnetic fields stimulation prevents steroid-induced osteonecrosis in rats. BMC Musculoskelet Disord 2011;12:215. https://doi.org/10.1186/1471-2474-12-215.
Zhou L, Yoon SJ, Jang KY, Moon YJ, Wagle S, Lee KB, et al. COMP-angiopoietin1 potentiates the effects of bone morphogenic protein-2 on ischemic necrosis of the femoral head in rats. PLoS One 2014;9(10):e110593. https://doi.org/10.1371/journal.pone.0110593.
Okazaki S, Nagoya S, Matsumoto H, Mizuo K, Shimizu J, Watanabe S, et al. TLR4 stimulation and corticosteroid interactively induce osteonecrosis of the femoral head in rat. J Orthop Res 2016;34(2):342-5. https://doi.org/10.1002/jor.23008.
Yu Z, Fan L, Li J, Ge Z, Dang X, Wang K. Lithium prevents rat steroid-related osteonecrosis of the femoral head by beta-catenin activation. Endocrine 2016;52(2):380-90. https://doi.org/10.1007/s12020-015-0747-y.
Zhang YL, Yin JH, Ding H, Zhang W, Zhang CQ, Gao YS. Vitamin K2 prevents glucocorticoid-induced osteonecrosis of the femoral head in rats. Int J Biol Sci 2016;12(4):347-58. https://doi.org/10.7150/ijbs.13269.
Tong PJ, Xu L, Hu BS, Jin HT, Li TY, Fang X. In vitro induction studies of YouGui drink on culture of steroid induced necrosis of femoral head rat osteoblast. [Article in Chinese]. Zhongguo Gu Shang 2010;23(1):23-7.
Chandra A, Lan S, Zhu J, Siclari VA, Qin L. Epidermal growth factor receptor (EGFR) signaling promotes proliferation and survival in osteoprogenitors by increasing early growth response 2 (EGR2) expression. J Biol Chem 2013;288(28):20488-98. https://doi.org/10.1074/jbc.M112.447250.
Bejar J, Peled E, Boss JH. Vasculature deprivation-induced osteonecrosis of the rat femoral head as a model for therapeutic trials. Theor Biol Med Model 2005;2:24. https://doi.org/10.1186/1742-4682-2-24.
Peled E, Bejar J, Barak M, Orion E, Norman D. Core decompression and alendronate treatment of the osteonecrotic rat femoral head: Computer-assisted analysis. Int J Exp Pathol 2013;94(3):212-6. https://doi.org/10.1111/iep.12022.
Peled E, Davis M, Axelman E, Norman D, Nadir Y. Heparanase role in the treatment of avascular necrosis of femur head. Thromb Res 2013;131(1):94-8. https://doi.org/10.1016/j.thromres.2012.09.018.
Terayama H, Ishikawa M, Yasunaga Y, Yamasaki T, Hamaki T, Asahara T, et al. Prevention of osteonecrosis by intravenous administration of human peripheral blood-derived CD34-positive cells in a rat osteonecrosis model. J Tissue Eng Regen Med 2011;5(1):32-40. https://doi.org/10.1002/term.285.
Vadasz Z, Misselevich I, Norman D, Peled E, Boss JH. Localization of vascular endothelial growth factor during the early reparative phase of the rats' vessels deprivation-induced osteonecrosis of the femoral heads. Exp Mol Pathol 2004;77(2):145-8. https://doi.org/10.1016/j.yexmp.2004.06.002.
Wang X, Chen W, Liu Q, Gao K, Wang G, Gao L, et al. Function and mechanism of mesoporous bioactive glass adsorbed epidermal growth factor for accelerating bone tissue regeneration. Biomed Mater 2017;12(2):025020. https://doi.org/10.1088/1748-605X/aa65d8.
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2018-03-18
Published 2018-11-07









