Simple, reliable, and time-efficient colorimetric method for the assessment of mitochondrial function and toxicity
DOI:
https://doi.org/10.17305/bjbms.2018.3323Keywords:
Janus green B, diethylsafranine, dehydrogenases, mitochondrial integrity, mitochondrial energizationAbstract
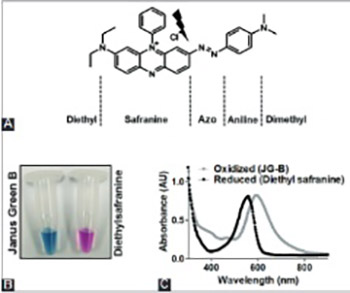
Mitochondria are organelles involved in the production of cellular energy, regulation of Ca2+ and redox signaling, and are critical for normal functioning of eukaryotic cells. The dysfunction of mitochondria has been implicated in a wide range of diseases, including metabolic and neurodegenerative disorders and different types of cancers. To better understand the role of mitochondria in healthy and disease states, the development of efficient and reliable tools for the assessment of mitochondrial function is particularly important. Janus green B (JG-B) is a supravital lipophilic cationic dye which, in its oxidized form, has a green-blue color. As JG-B is taken up and reduced by metabolically active mitochondria, the dye has been used for assessing the purity, integrity and metabolic activity of mitochondria with microscopy-based methods. Here we present a simple, time- and cost-efficient JG-B-based colorimetric assay for assessing mitochondrial function, activity and toxicity. The method is based upon reduction of JG-B by mitochondrial dehydrogenases to diethylsafranine, which is pink colored and has a maximum absorption at 550 nm. In this proof of principle study, using in vitro mitochondrial preparations isolated from rat brain, we provide evidence that monitoring JG-B conversion to diethylsafranine can be used as a reliable and robust indicator of mitochondrial activity and toxicity. Because of its simplicity and efficiency in terms of costs and time, this assay has a wide potential in analytical as well as therapeutic areas of biomedical research.
Citations
Downloads
References
Chen LB. Potential use of mitochondria as a reservoir for photosensitive lipophilic cations. New Dir Photodyn Ther 1987;847:46-50. https://doi.org/10.1117/12.942688.
Ross MF, Kelso GF, Blaikie FH, James AM, Cocheme HM, Filipovska A, et al. Lipophilic triphenylphosphonium cations as tools in mitochondrial bioenergetics and free radical biology. Biochemistry (Mosc) 2005;70(2):222-30. https://doi.org/10.1007/s10541-005-0104-5.
Todd AS, Barnetson WK. Use of dark ground microscopy in haematology. J Clin Pathol 1988;41(7):786-92. https://doi.org/10.1136/jcp.41.7.786.
Chaiyarit S, Thongboonkerd V. Changes in mitochondrial proteome of renal tubular cells induced by calcium oxalate monohydrate crystal adhesion and internalization are related to mitochondrial dysfunction. J Proteome Res 2012;11(6):3269-80. https://doi.org/10.1021/pr300018c.
Chen R, Liu W, Zhang G, Ye J. Mitochondrial proteomic analysis of cytoplasmic male sterility line and its maintainer in wheat (Triticum aestivum L.). Agric Sci China 2010;9(6):771-82. https://doi.org/10.1016/S1671-2927(09)60154-1.
Dutta M, Ghosh AK, Mohan V, Mishra P, Rangari V, Chattopadhyay A, et al. Antioxidant mechanism(s) of protective effects of Fenugreek 4-hydroxyisoleucine and trigonelline enriched fraction (TF4H (28%)) Sugaheal against copper-ascorbate induced injury to goat cardiac mitochondria in vitro. J Pharm Res 2014;8(6):798-811.
Onyia GOC, Gahan PB, Norman H. The use of new probes for protoplast integrity following isolation and purification of photoplasts from tubers of white yam (Discorea rotundata, poir). Plant Sci Lett 1984;33(2):231-8. https://doi.org/10.1016/0304-4211(84)90013-0.
Yu G, Xiang H, Tian J, Yin J, Pinkert CA, Li Q, et al. Mitochondrial haplotypes influence metabolic traits in porcine transmitochondrial cybrids. Sci Rep 2015;5:13118. https://doi.org/10.1038/srep13118.
Xiong H, Du S, Ni J, Zhou J, Yao J. Mitochondria and nuclei dual-targeted heterogeneous hydroxyapatite nanoparticles for enhancing therapeutic efficacy of doxorubicin. Biomaterials 2016;94:70-83. https://doi.org/10.1016/j.biomaterials.2016.04.004.
Xue T, Luo P, Zhu H, Zhao Y, Wu H, Gai R, et al. Oxidative stress is involved in Dasatinib-induced apoptosis in rat primary hepatocytes. Toxicol Appl Pharmacol 2012;261(3):280-91. https://doi.org/10.1016/j.taap.2012.04.010.
Ye Y, Huang A, Huang C, Liu J, Wang B, Lin K, et al. Comparative mitochondrial proteomic analysis of hepatocellular carcinoma from patients. Proteomics Clin Appl 2013;7(5-6):403-15. https://doi.org/10.1002/prca.201100103.
Yao X, Li M, He J, Zhang G, Wang M, Ma J, et al. Effect of early acute high concentrations of iodide exposure on mitochondrial superoxide production in FRTL cells. Free Radic Biol Med 2012;52(8):1343-52. https://doi.org/10.1016/j.freeradbiomed.2012.02.002.
Ma WW, Hou CC, Zhou X, Yu HL, Xi YD, Ding J, et al. Genistein alleviates the mitochondria-targeted DNA damage induced by beta-amyloid peptides 25-35 in C6 glioma cells. Neurochem Res 2013;38(7):1315-23. https://doi.org/10.1007/s11064-013-1019-y.
Stephanova E, Topouzova-Hristova T, Konakchieva R. Mitochondria are involved in stress response of A549 alveolar cells to halothane toxicity. Toxicol Vitr 2008;22(3):688-94. https://doi.org/10.1016/j.tiv.2007.12.012.
Du Buy HG, Showacre J. Enzymes catalyzing sequential reactions in mouse brain and liver supernatant fractions: I. Differential use of Janus green B and phenazine methosulfate. J Histochem Cytochem 1959;7(6):361-9. https://doi.org/10.1177/7.6.361.
Lazarow A, Cooperstein S. Studies on the enzymatic basis for the Janus green B staining reaction. J Histochem Cytochem 1953;1(4):234-41. https://doi.org/10.1177/1.4.234.
Tanaka Y. Deposition of Janus green B and pinocyanol in mitochondria of supravitally stained KB cells. Exp Cell Res 1968;52(2):338-48.
https://doi.org/10.1016/0014-4827(68)90475-8.
Showacre JL. A critical study of Janus Green B coloration as a tool for characterizing mitochondria. J Natl Cancer Inst 1953;13(4):829-45.
Udhayabanu T, Manole A, Rajeshwari M, Varalakshmi P, Houlden H, Ashokkumar B. Riboflavin responsive mitochondrial dysfunction in neurodegenerative diseases. J Clin Med 2017;6(5). pii: E52. https://doi.org/10.3390/jcm6050052.
Ghazi-Khansari M, Mohammadi-Bardbori A, Hosseini MJ. Using Janus green B to study paraquat toxicity in rat liver mitochondria: Role of ACE inhibitors (thiol and nonthiol ACEi). Ann N Y Acad Sci 2006;1090:98-107. https://doi.org/10.1196/annals.1378.010.
Mohammadi-Bardbori A, Ghazi-Khansari M. Comparative measurement of cyanide and paraquat mitochondrial toxicity using two different mitochondrial toxicity assays. Toxicol Mech Methods 2007;17(2):87-91. https://doi.org/10.1080/15376510600822664.
Cooperstein SJ, Lazarow A, Patterson JW. II. Reactions and properties of Janus green B and its derivatives. Exp Cell Res 1953;5(1):69-82. https://doi.org/10.1016/0014-4827(53)90095-0.
Perry SW, Norman JP, Barbieri J, Brown EB, Gelbard HA. Mitochondrial membrane potential probes and the proton gradient: A practical usage guide. Biotechniques 2011;50(2):98-115. https://doi.org/10.2144/000113610.
Cottet-Rousselle C, Ronot X, Leverve X, Mayol JF. Cytometric assessment of mitochondria using fluorescent probes. Cytometry A 2011;79(6):405-25. https://doi.org/10.1002/cyto.a.21061.
Hamberger A, Blomstrand C, Lehninger AL. Comparative studies on mitochondria isolated from neuron enriched and glia enriched fractions of rabbit and beef brain. J Cell Biol 1970;45(2):221-34. https://doi.org/10.1083/jcb.45.2.221.
Dua R, Gill KD. Effect of aluminium phosphide exposure on kinetic properties of cytochrome oxidase and mitochondrial energy metabolism in rat brain. Biochim Biophys Acta 2004;1674(1):4-11. https://doi.org/10.1016/j.bbagen.2004.05.003.
Valenti D, Vacca RA, de Pinto MC, De Gara L, Marra E, Passarella S. In the early phase of programmed cell death in Tobacco Bright Yellow 2 cells the mitochondrial adenine nucleotide translocator, adenylate kinase and nucleoside diphosphate kinase are impaired in a reactive oxygen species-dependent manner. Biochim Biophys Acta 2007;1767(1):66-78. https://doi.org/10.1016/j.bbabio.2006.11.004.
Cooperstein SJ, Dixit PK, Lazarow A. Studies on the mechanism of Janus green B staining of mitochondria. IV. Reduction of Janus green B by isolated cell fractions. Anat Rec 1960;138(1):49-66. https://doi.org/10.1002/ar.1091380107.
Lazarow A, Cooperstein SJ. Studies on the enzymatic basis for the Janus Green B staining reaction. J Histochem Cytochem 1953;1(4):234-41. https://doi.org/10.1177/1.4.234.
Braun S, Erdelyi M, Udvardy A. Janus Green B and the biologic “oxygen effect''. Cancer Res 1967;27(4):660-7.
Koide T, Baba K, Wa K, Keda M. Mitochondrial injury produced by Janus Green B - Enzyme-morphological and ultrastructural observation. Acta Histochem Cytochem 1971;4(3):137-52. https://doi.org/10.1267/ahc.4.137.
Johnson LV, Walsh ML, Chen B, Buchanan JM. Localization of mitochondria in living cells with rhodamine 123. Proc Natl Acad Sc USA Cell Biol 1980;77(2):990-4. https://doi.org/10.1073/pnas.77.2.990.
Chen Y, Qiao L, Yu B, Li G, Liu C, Ji L, et al. Mitochondria-specific phosphorescent imaging and tracking in living cells with an AIPE-active iridium(III) complex. Chem Commun (Camb) 2013;49(94):11095-7. https://doi.org/10.1039/c3cc46957c.
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2018-05-14
Published 2018-11-07









