Formulation ingredients for toothpastes and mouthwashes
DOI:
https://doi.org/10.17305/bjbms.2004.3362Keywords:
formulation, ingredients, toothpastes, mouthwashesAbstract
In order to achieve the multi-claim products required for the dental care category, it is necessary for the formulator to use a variety of different ingredients. This places a number of demands on the development process. Innovations in the areas of pharmaceutical technology have contributed to the formulation of the products having superior efficacy as well as other attributes that may contribute to clinical response and patient acceptability. Improved clinical efficacy and tolerability, along with conditioning signals, should encourage patient compliance with oral hygiene further complementing professional efforts directed at disease prevention. The most effective way of preventing the development of dental disease is in controlling the production of dental plaque. It is formed by microbial action. The removal of plaque from the teeth and related areas is essential for the maintenance of a healthy mouth.
In this paper we have presented the main components of toothpastes and mouthwashes. For the active ingredients, their supposed effect as therapeutic agents is also explained.
Citations
Downloads
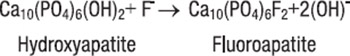
Published
How to Cite
Accepted 2018-03-14
Published 2004-11-20









