Targeted immunotherapy with a checkpoint inhibitor in combination with chemotherapy: A new clinical paradigm in the treatment of triple-negative breast cancer
DOI:
https://doi.org/10.17305/bjbms.2019.4204Keywords:
Breast cancer, triple-negative breast cancer, immune checkpoint inhibitors, predictive biomarkers, PD-L1Abstract
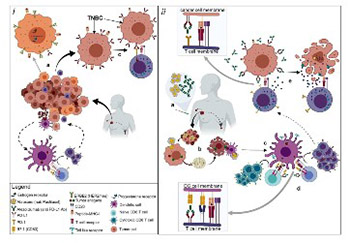
The treatment of several solid and hematologic malignancies with immune checkpoint inhibitors (against programmed death receptor-1/ligand-1 [PD-1/PD-L1]) has dramatically changed the cancer treatment paradigm. However, no checkpoint inhibitors were previously approved for the treatment of triple-negative breast cancer (TNBC), a difficult-to-treat disease with a high unmet therapeutic need. Based on IMpassion130 clinical trial (NCT02425891), the Food and Drug Administration (FDA) has recently granted an accelerated approval for atezolizumab (TECENTRIQ®), a monoclonal antibody drug targeting PD-L1, plus chemotherapy (Abraxane; nab®-Paclitaxel) for the treatment of adults with PD-L1-positive, unresectable, locally advanced or metastatic TNBC. The FDA has also approved the Ventana diagnostic antibody SP142 as a companion test for selecting TNBC patients for treatment with atezolizumab. In the present review, we briefly discuss the importance of this breakthrough as the first cancer immunotherapy regimen to be approved for the management of breast cancer.
Citations
Downloads