The value of lymphocyte count in determining the severity of COVID-19 and estimating the time for nucleic acid test results to turn negative
DOI:
https://doi.org/10.17305/bjbms.2020.4868Keywords:
COVID-19, lymphocyte count, SARS-CoV-2 nucleic acid testAbstract
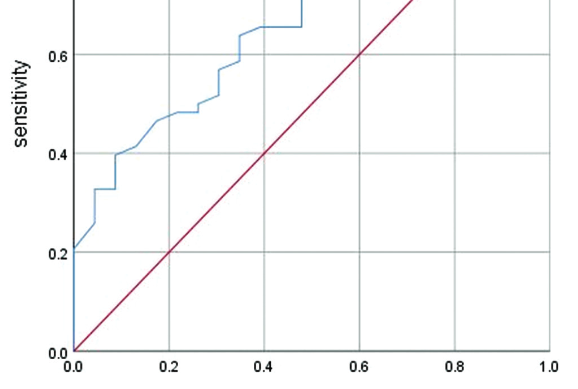
Peripheral blood lymphocyte count is shown to be decreased in patients with COVID-19 in the early stage of the disease. The degree of lymphocyte count reduction is related to COVID-19 severity and could be used as an indicator to reflect the disease severity. Our aim was to investigate the value of lymphocyte count in determining COVID-19 severity and estimating the time for SARS-CoV-2 nucleic acid test results to turn negative. We retrospectively analyzed clinical data of 201 patients with severe and critical COVID-19. The patients were admitted to the West Campus of Union Hospital of Tongji Medical College of Huazhong University of Science and Technology. The data included age, gender, chronic disease, lymphocyte count, and SARS-CoV-2 nucleic acid test results. The age of patients in critically ill group was higher than in severely ill group (p = 0.019). The lymphocyte count of critically ill patients was lower than of severely ill patients. The cutoff value of lymphocyte count to distinguish between the critically ill and the severely ill was 0.735 × 109/L (p = 0.001). The cutoff value of lymphocyte count for SARS-CoV-2 nucleic acid test results turning negative in severely and critically ill patients with chronic diseases (hypertension, diabetes, and coronary heart disease) was 0.835 × 109/L (p = 0.017). The cutoff value of lymphocyte count for SARS-CoV-2 nucleic acid test results turning negative in severely and critically ill male patients was 0.835 × 109/L (p < 0.0001). Lymphocyte count could be an effective indicator to predict COVID-19 severity. It may also be useful in determining the time for nucleic acid test results to turn negative in COVID-19 patients with underlying chronic diseases or male COVID-19 patients with severe and critical conditions.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2020 Yuanchao Li, Tuoyun Yang, Sicong Wang, Junbo Zheng, Jing Zhou, Min Jiang, Tong Zhou, Yang Cao, Hongliang Wang

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2020-08-24
Published 2021-04-01









