Efficacy of chemotherapeutics in classic and non-classic Kaposi sarcoma: A single-center retrospective real-world data
DOI:
https://doi.org/10.17305/bjbms.2020.5329Keywords:
Kaposi sarcoma, chemotherapy, pegylated liposomal doxorubicin, paclitaxelAbstract
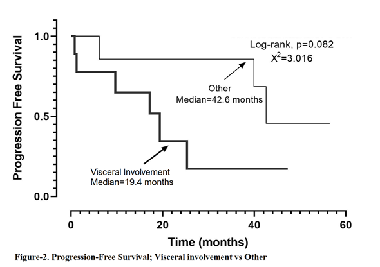
Kaposi sarcoma is a rare disease and there is a gap in the literature about which chemotherapeutics should be applied, especially for the classical type. We aimed to present our institutional data on the demographic characteristics, treatment, and treatment efficacy in 16 Kaposi sarcoma (KS) patients treated with chemotherapy. We retrospectively analyzed the demographic and clinical characteristics, and the chemotherapeutic agents administered to the 16 KS patients diagnosed in our center and treated with chemotherapy, based on the medical records obtained. The median age, gender, type of KS, site of involvement, cytotoxic agents administered, progression-free survival (PFS), overall survival (OS), objective response rate (ORR), disease control rate (DCR), and safety profiles of the patients were evaluated. The median age at disease onset was 61.07 years (range, 39.4–85.8 years). Among the patients, 1 had immunosuppression-related KS, 4 had AIDS-related KS, and 11 had classical KS. In the first-line cytotoxic therapy, 7 patients received pegylated-liposomal doxorubicin (PLD), 6 patients received paclitaxel, 2 patients received oral etoposide, and 1 patient received the adriamycin, bleomycin, and vincristine regimen. In the Kaplan–Meier analysis, the PFS was 39.9 months (95% CI, 7.7–72.0). In the first-line setting, a significant difference in terms of PFS was observed between the PLD- and paclitaxel-treated groups (not reached vs. 12.8 months, p = 0.033). The OS was 66.1 months (95% CI, 30.2–102.0). The ORR of the 16 patients was 43.8%, and their DCR was 81.3%. No grade 3 or 4 toxicity was observed. This retrospective study showed that PLD seems better than paclitaxel in terms of PFS and response rates and it has shown to have a good safety profile in KS patients.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2021-02-07
Published 2021-12-01









