SARS-CoV-2 infection: Understanding the immune system abnormalities to get an adequate diagnosis
DOI:
https://doi.org/10.17305/bjbms.2020.5400Keywords:
SARS-CoV-2, diagnosis, macrophage, lymphocyte, T-cells, inflammation, COVID-19Abstract
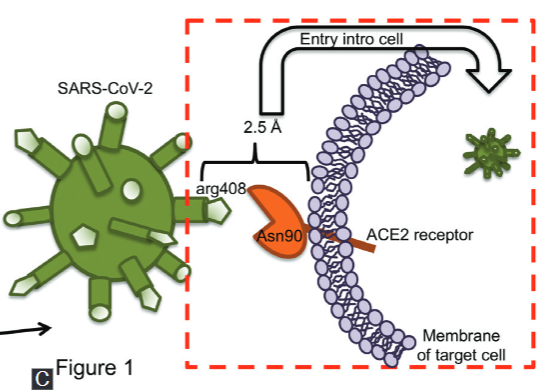
COVID-19 is the current pandemic caused by the novel coronavirus, SARS-CoV-2, that emerged from China at the end of December 2019. The scientific community is making extraordinary efforts to understand the virus structure and the pathophysiology and immunological processes activated in the host, in order to identify biomarkers, diagnostic tools, treatments, and vaccines to decrease COVID-19 incidence and mortality. Various abnormalities have been noted during SARS-CoV-2 infection both in lymphoid and myeloid cells. Such abnormalities may disturb the immune system function and cause a massive inflammatory response that impairs tissue function. This review discusses the close relationship between the immune system abnormalities and the broad spectrum of clinical manifestations, including fibrosis, in the context of COVID-19 disease. Moreover, we described the current strategies for COVID-19 diagnosis, and we provide a summary of the most useful clinical laboratory parameters to identify severe COVID-19 patients.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2021 Karen Medina-Quero, Omar Barreto-Rodriguez, Voltaire Mendez-Rodriguez, Anahí Sanchez-Moncivais, Ivette Buendia-Roldan, Leslie Chavez-Galan

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-02-12
Published 2021-10-01









