Construction and verification of prognostic nomogram for early-onset esophageal cancer
DOI:
https://doi.org/10.17305/bjbms.2021.5533Keywords:
Early-onset esophageal cancer (EOEC), nomogram, overall survival (OS), cancer-specific survival (CSS), Surveillance, Epidemiology, and End Results (SEER)Abstract
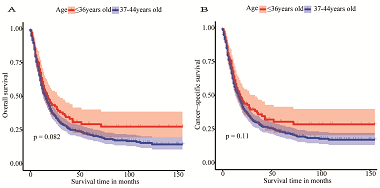
This study aimed to build up nomogram models to evaluate overall survival (OS) and cancer-specific survival (CSS) in early-onset esophageal cancer (EOEC). Patients diagnosed with esophageal cancer (EC) from 2004 to 2015 were extracted from the Surveillance Epidemiology and End Results (SEER) database. Clinicopathological characteristics of younger versus older patients were compared, and survival analysis was performed in both groups. Independent related factors influencing the prognosis of EOEC were identified by univariate and multivariate Cox analysis, which were incorporated to construct a nomogram. The predictive capability of the nomogram was estimated by the concordance index (C-index), calibration plot, receiver operating characteristic (ROC) curve, and decision curve analysis (DCA). A total of 534 younger and 17,243 older patients were available from the SEER database. Younger patients were randomly segmented into a training set (n = 266) and a validation set (n = 268). In terms of the training set, the C-index of the OS nomogram was 0.740 (95% CI: 0.707-0.773), and that of the CSS nomogram was 0.752 (95% CI: 0.719-0.785). In view of the validation set, the C-index of OS and CSS were 0.706 (95% CI: 0.671-0.741) and 0.723 (95% CI: 0.690-0.756), respectively. Calibration curves demonstrated the consistent degree of fit between actual and predicted values in nomogram models. From the perspective of DCA, the nomogram models were more beneficial than the tumor-node-metastasis (TNM) and the SEER stage for EOEC. In brief, the nomogram model can be considered as an individualized quantitative tool to predict the prognosis of EOEC patients to assist clinicians in making treatment decisions.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2021 Xiaoxiao Liu, Wei Guo, Xiaobo Shi, Yue Ke, Yuxing Li; Shupei Pan, Yingying Jin, Yuchen Wang, Qinli Ruan; Hongbing Ma

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-03-14
Published 2021-12-01









