Poorly differentiated clusters and tumor budding are important prognostic factors in colorectal carcinomas
DOI:
https://doi.org/10.17305/bjbms.2021.6110Keywords:
Colorectal carcinomas, tumor budding, poorly differentiated clusters, grading system, prognostic factorsAbstract
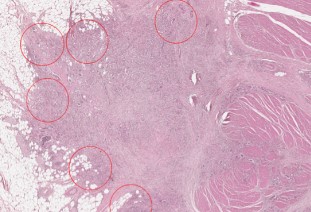
The aim of our study was to assess the prognostic value of the two new grading systems based on the quantification of tumor budding - TB (GBd) and poorly differentiated clusters - PDCs (PDCs-G) in colorectal carcinomas (CRC). We performed a retrospective study on 71 CRC patients who underwent surgery at the Emergency County Hospital, Timișoara. CRC cases were classified based on haematoxylin-eosin slides, using the conventional grading system, GBd and PDCs-G, respectively. We used two-tier and three-tier grading schemes for each system. Subsequently, we evaluated associations with other prognostic factors in CRC. Based on the three-tier GBd (GBd-3t) most cases (34/69, 49.27%) were classified as G3Bd-3t, while based on the conventional grading system, the majority of the cases (55/69, 79.71%) were considered G2. On the other hand, based on the three-tier PDCs-G system (PDCs-G-3t), most cases (31/69, 44.93%) were PDCs-G2-3t. We also noted a more significant association of GBd-3t with other prognostic parameters analyzed, as compared to the conventional grading system. Nodal status, tumor stage, and lymphovascular invasion were strongly correlated with GBd-3t (p=0.0001). Furthermore, we noted that PDCs-G-3t correlated more significantly than the conventional grading system with nodal status (p<0.0001), tumor stage (p=0.0003), lymphovascular invasion (p<0.0001), perineural invasion (p=0.005) and the tumor border configuration (p<0.0001). High GBd and PDCs-G grades correlate directly with other negative prognostic factors in CRC.Thus, these new parameters/classification methods could be used as additional tools for risk stratification in patients with CRC.
Citations
Downloads
Downloads
Additional Files
Published
License
Copyright (c) 2021 Aura Jurescu, Adrian Văduva, Adelina Gheju

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-08-27
Published 2022-04-01









