Sustained seroprevalence of SARS-CoV-2 antibodies one year after infection: one of the first COVID-19 cluster cases in Bosnia and Herzegovina
DOI:
https://doi.org/10.17305/bjbms.2021.6340Keywords:
Bosnia and Herzegoina, pandemic, SARS-CoV-2, COVID-19, immune response, seroconversion, IgGAbstract
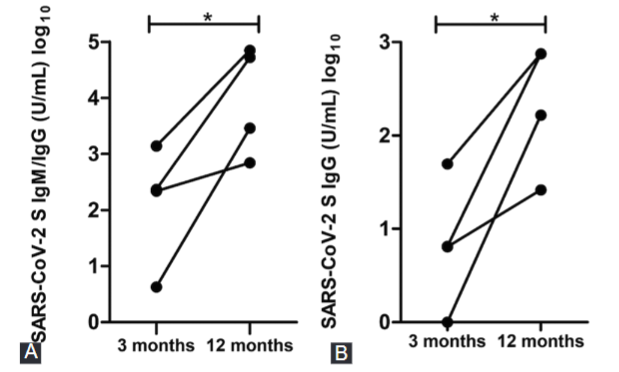
SARS-CoV-2 (severe acute respiratory syndrome coronavirus type 2) is a novel virus that has been identified as a causal agent of COVID-19, an emergent infectious disease which brought about a new pandemic in the twenty-first century. The immune responses and clinical features of individuals infected with SARS-CoV-2 have not yet been fully described. Thus, in this study, we compare the seroprevalence and define the correlation between symptoms and serological results in the first COVID-19 cluster in the city of Konjic, Bosnia and Herzegovina. Of the total number, 93% of RT-PCR positive participants had positive IgG serology and 75% of them developed symptoms of COVID-19. We found that there was no significant alteration in specific IgG (p = 0.504) antibody levels during the 1-year period after COVID-19. Our results indicate that symptomatic COVID-19 patients have a higher rate of seroconversion (p < 0.01). The IgG seroconversion was correlated with high fever (p = 0.002) and headache (p = 0.007), suggesting that these symptoms could be considered as indicators of a better immune response. This study has demonstrated persistence of sustained levels of specific SARS-CoV-2 antibodies after recovering from COVID-19 infection. However, in order to gain a better insight into the immune response to SARS-CoV-2, further systematic studies should be focused on quality and longevity analyses.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2021 Božo Šušak, Vinka Mikulić, Armina Lazarević, Ivanka Mikulić, Jurica Arapovic

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-08-08
Published 2022-02-01









