Clinical attachment loss in the use of adjunctive antimicrobial photodynamic therapy in Stages II-IV Grade C molar-incisor periodontitis: A systematic review and meta-analysis
DOI:
https://doi.org/10.17305/bjbms.2022.7157Keywords:
Aggressive periodontitis, Attachment loss, Photo chemotherapy, Periodontal debridement, Dental scaling, TherapyAbstract
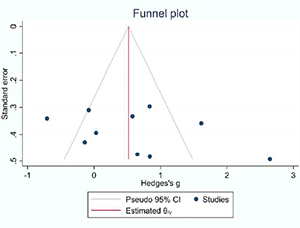
This systematic review and meta-analysis aimed to assess the extent of clinical attachment loss (CAL) as a clinical parameter in the efficacy of antimicrobial photodynamic therapy (aPDT) in non-surgical management of stage II-IV grade C molar-incisor pattern Periodontitis. This review protocol was conducted in accordance with PRISMA statements and is registered in PROSPERO (CRD42022321211). An electronic and manual search was conducted for relevant articles comparing the efficacy of aPDT versus scaling and root planning (SRP) alone or with amoxicillin/metronidazole (AMX/MET) published up until December 2021. The mean clinical attachment loss (CAL), probing depth (PD) reduction, and bleeding on probing (BOP) with a 95% confidence interval (CI) were pooled and compared between the two groups with CAL < and > 7 mm using a random-effect model after 3 and 6 months. To assess the heterogeneity of the findings, the I2 test was applied and Publication bias was evaluated by visual examination of the funnel plot symmetry. Analysis of 9 studies indicated a significant difference in clinical attachment gain in patients with CAL > 7 mm between the aPDT group and the SRP alone (mean difference=0.92, 95% CI=0.01-1.84, P=0.05) and SRP + AMX/MET (mean difference=0.91, 95% CI=-0.14-1.68, P=0.02) control groups. However, this difference was not significant in patients with CAL < 7 mm. Despite the limitations of the included studies, aPDT can be suggested for the improvement of clinical parameters in grade C molar-incisor pattern Periodontitis with CAL > 7 mm. However, its application in milder cases requires further investigation.
Citations
Downloads
Downloads
Additional Files
Published
License
Copyright (c) 2022 Zahra Baghani, Samira Basir Shabestari, Malihe Karrabi

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2022-05-02
Published 2022-10-23









