Effect of met-enkephalin on chromosomal aberrations in the lymphocytes of the peripheral blood of patients with multiple sclerosis
DOI:
https://doi.org/10.17305/bjbms.2014.2267Keywords:
met-enkephalin, chromosomal aberrations, multiple sclerosis, peripheral blood lymphocytesAbstract
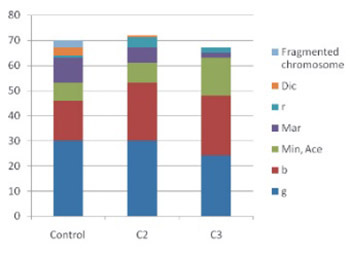
Endogenious opiod met-enkephalin throughout previous research manifested cytoprotective and anti-inflammatory effects. Previous research suggests that met-enkephalin has cytogenetic effects. Reducement in the frequency of structural chromosome aberrations as well as a suppressive effect on lymphocyte cell cycle is found. It also reduces apoptosis in the blood samples of the patients with immune-mediated diseases. Met-enkephalin exerts immunomodulatory properties and induces stabilization of the clinical condition in patients with multiple Sclerosis (MS). The goal of the present research was to evaluate met-enkephalin in vitro effects on the number and type of chromosome aberrations in the peripheral blood lymphocytes of patients with MS. Our research detected disappearance of ring chromosomes and chromosome fragmentations in the cultures of the peripheral blood lymphocytes treated with met-enkephalin (1.2 μg/mL). However, this research did not detect any significant effects of met-enkephalin on the reduction of structural chromosome aberrations and disappearance of dicentric chromosomes. Chromosomes with the greatest percent of inclusion in chromosome aberrations were noted as: chromosome 1, chromosome 2 and chromosome 9. Additionally, we confirmed chromosome 14 as the most frequently included in translocations. Furthermore, met-enkephalin effects on the increase of the numerical aberrations in both concentrations applied were detected. Those findings should be interpreted cautiously and more research in this field should be conducted.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-07-12
Published 2014-05-20









