The utility of procollagen type 1 N-terminal propeptide for the bone status assessment in postmenopausal women
DOI:
https://doi.org/10.17305/bjbms.2013.2337Keywords:
procollagen type 1 N-terminal propeptide, postmenopausal females, osteoporosis, bone mineral densityAbstract
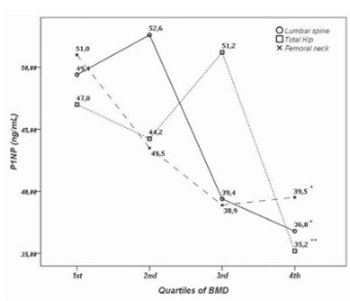
The utility of procollagen type 1 N-terminal propeptide (P1NP) in the management of metabolic bone diseases remains a subject of debate since the reference ranges are not rigorously established and fail to account for many of the preanalytical variables. We aimed to establish reference intervals for P1NP level in healthy and osteoporotic postmenopausal females stratified by age, body mass index and menopausal duration. We also aimed to assess the relationship between P1NP and BMD. This cross-sectional study enrolled 183 postmenopausal females who were divided in osteoporosis group (N=93) and control group (N=90) with preserved bone mass based on BMD assessed by DXA. In the osteoporosis group median P1NP was significantly higher (51.7 ng / mL; 95%CI 43.2-53.7) compared to control group (38.9 ng/mL; 95%CI 34.2-43.9)(p<0.01). After controlling for age, BMI and years since menopause, there was significant inverse association between BMD and P1NP at the femoral neck (r=-0.18), total hip (r=-0.207) and lumbar spine (r=-0.236). There was no significant difference in P1NP concentration across quartiles of age in postmenopausal females. P1NP was significantly lower in obese postmenopausal females with preserved bone mass compared to normal weight and overweight females in control and in osteoporosis group.
In conclusion, we showed that P1NP is inversely associated with BMD even after controlling for age, BMI and years since menopause. Although, P1NP is significantly higher in postmenopausal females with osteoporosis compared to postmenopausal females with preserved bone mass its low specificity does not warrant its utility is diagnosing osteoporosis.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
How to Cite
Accepted 2017-08-08
Published 2013-11-20









