Matrine induced G0/G1 arrest and apoptosis in human acute T-cell lymphoblastic leukemia (T-ALL) cells
DOI:
https://doi.org/10.17305/bjbms.2017.2457Keywords:
Sophora flavescens, matrine, acute lymphoblastic leukemia, T-ALL, CCRF-CEM, miR-376b, miR-106b, p21, CDKN1A, cell cycle, G0/G1 arrest, apoptosis, autophagyAbstract
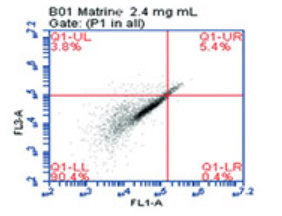
Matrine, a natural product extracted from the root of Sophora flavescens, is a promising alternative drug in different types of cancer. Here, we aimed to investigate the therapeutic effects and underlying molecular mechanisms of matrine on human acute lymphoblastic leukemia (ALL) cell line, CCRF-CEM. Cell viability and IC50 values were determined by WST-1 cell cytotoxicity assay. Cell cycle distribution and apoptosis rates were analyzed by flow cytometry. Expression patterns of 44 selected miRNAs and 44 RNAs were analyzed by quantitative reverse transcription polymerase chain reaction (qRT-PCR) using the Applied Biosystems 7500 Fast Real-Time PCR System. Matrine inhibited cell viability and induced apoptosis of CCRF-CEM cells in a dose-dependent manner. Cell cycle analysis demonstrated that matrine-treated CCRF-CEM cells significantly accumulated in the G0/G1 phase compared with the untreated control cells. hsa-miR-376b-3p (-37.09 fold, p = 0.008) and hsa-miR-106b-3p (-16.67 fold, p = 0.028) expressions were decreased, whereas IL6 (95.47 fold, p = 0.000011) and CDKN1A (140.03 fold, p = 0.000159) expressions were increased after matrine treatment. Our results suggest that the downregulation of hsa-miR-106b-3p leads to the upregulation of target p21 gene, CDKN1A, and plays a critical role in the cell cycle progression by arresting matrine-treated cells in the G0/G1 phase.
Citations
Downloads
References
Evangelisti C, Evangelisti C, Chiarini F, Lonetti A, Buontempo F, Neri LM, et al. Autophagy in acute leukemias: A double-edged sword with important therapeutic implications. Biochim Biophys Acta 2015;1853(1):14-26. https://doi.org/10.1016/j.bbamcr.2014.09.023.
Richard NP, Pippa R, Cleary MM, Puri A, Tibbitts D, Mahmood S, et al. Combined targeting of SET and tyrosine kinases provides an effective therapeutic approach in human T-cell acute lymphoblastic leukemia. Oncotarget 2016;7(51):84214-27. https://doi.org/10.18632/oncotarget.12394.
Jovanovic M, Hengartner MO. MiRNAs and apoptosis: RNAs to die for. Oncogene 2006;25(46):6176-87. https://doi.org/10.1038/sj.onc.1209912.
Jing Z, Han W, Sui X, Xie J, Pan H. Interaction of autophagy with microRNAs and their potential therapeutic implications in human cancers. Cancer Lett 2015;356(2B):332-8. https://doi.org/10.1016/j.canlet.2014.09.039.
Alenzi FQ. Links between apoptosis, proliferation and the cell cycle. Br J Biomed Sci 2004;61(2):99-102. https://doi.org/10.1080/09674845.2004.11732652.
Wang RC, Levine B. Autophagy in cellular growth control. FEBS Lett 2010;584(7):1417-26. https://doi.org/10.1016/j.febslet.2010.01.009.
Eisenberg-Lerner A, Bialik S, Simon HU, Kimchi A. Life and death partners: Apoptosis, autophagy and the cross-talk between them. Cell Death Differ 2009;16(7):966-75. https://doi.org/10.1038/cdd.2009.33.
Noguchi M, Hirata N, Edamura T, Ishigaki S, Suizu F. Intersection of apoptosis and autophagy cell death pathways. Austin J Mol Cell Biol 2015;2(2):1004.
Johnson CD, Esquela-Kerscher A, Stefani G, Byrom M, Kelnar K, Ovcharenko D, et al. The let-7 microRNA represses cell proliferation pathways in human cells. Cancer Res 2007;67(16):7713-22. https://doi.org/10.1158/0008-5472.CAN-07-1083.
Calin GA, Cimmino A, Fabbri M, Ferracin M, Wojcik SE, Shimizu M, et al. MiR-15a and miR-16-1 cluster functions in human leukemia. Proc Natl Acad Sci U S A 2008;105(13):5166-71. https://doi.org/10.1073/pnas.0800121105.
Linsley PS, Schelter J, Burchard J, Kibukawa M, Martin MM, Bartz SR, et al. Transcripts targeted by the microRNA-16 family cooperatively regulate cell cycle progression. Mol Cell Biol 2007;27(6):2240-52. https://doi.org/10.1128/MCB.02005-06.
Ivanovska I, Ball AS, Diaz RL, Magnus JF, Kibukawa M, Schelter JM, et al. MicroRNAs in the miR-106b family regulate p21/CDKN1A and promote cell cycle progression. Mol Cell Biol 2008;28(7):2167-74. https://doi.org/10.1128/MCB.01977-07.
Gillies JK, Lorimer IA. Regulation of p27Kip1 by miRNA 221/222 in glioblastoma. Cell Cycle 2007;6(16):2005-9. https://doi.org/10.4161/cc.6.16.4526.
Lujambio A, Esteller M. CpG island hypermethylation of tumor suppressor microRNAs in human cancer. Cell Cycle 2007;6(12):1455-9. https://doi.org/10.4161/cc.6.12.4408.
Yu Z, Baserga R, Chen L, Wang C, Lisanti MP, Pestell RG. MicroRNA, cell cycle, and human breast cancer. Am J Pathol 2010;176(3):1058-64. https://doi.org/10.2353/ajpath.2010.090664.
le Sage C, Nagel R, Egan DA, Schrier M, Mesman E, Mangiola A, et al. Regulation of the p27(Kip1) tumor suppressor by miR-221 and miR-222 promotes cancer cell proliferation. EMBO J 2007;26(15):3699-708. https://doi.org/10.1038/sj.emboj.7601790.
Cimmino A, Calin GA, Fabbri M, Iorio MV, Ferracin M, Shimizu M, et al. MiR-15 and miR-16 induce apoptosis by targeting BCL2. Proc Natl Acad Sci U S A 2005;102(39):13944-9. https://doi.org/10.1073/pnas.0506654102.
Tusong H, Maolakuerban N, Guan J, Rexiati M, Wang WG, Azhati B, et al. Functional analysis of serum microRNAs miR-21 and miR-106a in renal cell carcinoma. Cancer Biomark 2017;18(1):79-85. https://doi.org/10.3233/CBM-160676.
He QY, Wang GC, Zhang H, Tong DK, Ding C, Liu K, et al. MiR-106a-5p suppresses the proliferation, migration, and invasion of osteosarcoma cells by targeting HMGA2. DNA Cell Biol 2016;35(9):506-20. https://doi.org/10.1089/dna.2015.3121.
Song J, Yin J, Bai Z, Zhang J, Meng H, Cai J, et al. The profile of serum microRNAs predicts prognosis for resected gastric cancer patients receiving platinum-based chemotherapy. Dig Dis Sci 2017;62(5):1223-34. https://doi.org/10.1007/s10620-017-4513-2.
Fan L, Qi H, Teng J, Su B, Chen H, Wang C, et al. Identification of serum miRNAs by nano-quantum dots microarray as diagnostic biomarkers for early detection of non-small cell lung cancer. Tumour Biol 2016;37(6):7777-84. https://doi.org/10.1007/s13277-015-4608-3.
Zhang GJ, Li JS, Zhou H, Xiao H, Li Y, Zhou T, et al. MicroRNA-106b promotes colorectal cancer cell migration and invasion by directly targeting DLC1. J Exp Clin Cancer Res 2015;34(1):73. https://doi.org/10.1186/s13046-015-0189-7.
Hu Y, Li K, Asaduzzaman M, Cuella R, Shi H, Raguz S, et al. MiR-106b~25 cluster regulates multidrug resistance in an ABC transporter-independent manner via downregulation of EP300. Oncol Rep 2016;35(2):1170-8. https://doi.org/10.3892/or.2015.4412.
Liang H, Studach L, Hullinger RL, Xie J, Andrisani OM. Down-regulation of RE-1 silencing transcription factor (REST) in advanced prostate cancer by hypoxia-induced miR-106b~25. Exp Cell Res 2014;320(2):188-99. https://doi.org/10.1016/j.yexcr.2013.09.020.
Fontana L, Fiori ME, Albini S, Cifaldi L, Giovinazzi S, Forloni M, et al. Antagomir-17-5p abolishes the growth of therapy-resistant neuroblastoma through p21 and BIM. PLoS One 2008;3(5):e2236. https://doi.org/10.1371/journal.pone.0002236.
Dutto I, Tillhon M, Cazzalini O, Stivala LA, Prosperi E. Biology of the cell cycle inhibitor p21CDKN1A: Molecular mechanisms and relevance in chemical toxicology. Arch Toxicol 2015;89(2):155-78. https://doi.org/10.1007/s00204-014-1430-4.
Kim YK, Yu J, Han TS, Park SY, Namkoong B, Kim DH, et al. Functional links between clustered microRNAs: Suppression of cell-cycle inhibitors by microRNA clusters in gastric cancer. Nucleic Acids Res 2009;37(5):1672-81. https://doi.org/10.1093/nar/gkp002.
Petrocca F, Visone R, Onelli MR, Shah MH, Nicoloso MS, de Martino I, et al. E2F1-regulated MicroRNAs ımpair TGFβ-dependent cell-cycle arrest and apoptosis in gastric cancer. Cancer Cell 2008;13(3):272-86. https://doi.org/10.1016/j.ccr.2008.02.013.
Wang Q, Du H, Geng G, Zhou H, Xu M, Cao H, et al. Matrine inhibits proliferation and induces apoptosis via BID-mediated mitochondrial pathway in esophageal cancer cells. Mol Biol Rep 2014;41(5):3009-20. https://doi.org/10.1007/s11033-014-3160-3.
Zhang LP, Jiang JK, Tam JW, Zhang Y, Liu XS, Xu XR, et al. Effects of matrine on proliferation and differentiation in K-562 cells. Leuk Res 2001;25(9):793-800. https://doi.org/10.1016/S0145-2126(00)00145-4.
Hu MJ, Zeng H, Wu YL, Zhang YP, Zhang S, Qiao MM, et al. Synergistic effects of matrine and 5-fluorouracil on tumor growth of the implanted gastric cancer in nude mice. Chin J Dig Dis 2005;6(2):68-71. https://doi.org/10.1111/j.1443-9573.2005.00201.x.
Liu XY, Fang H, Yang ZG, Wang XY, Ruan LM, Fang DR, et al. Matrine inhibits invasiveness and metastasis of human malignant melanoma cell line A375 in vitro. Int J Dermatol 2008;47(5):448-56. https://doi.org/10.1111/j.1365-4632.2008.03627.x.
Ma L, Wen S, Zhan Y, He Y, Liu X, Jiang J. Anticancer effects of the Chinese medicine matrine on murine hepatocellular carcinoma cells. Planta Med 2008;74(3):245-51. https://doi.org/10.1055/s-2008-1034304.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001;25(4):402-8. https://doi.org/10.1006/meth.2001.1262.
Niu H, Zhang Y, Wu B, Zhang Y, Jiang H, He P. Matrine induces the apoptosis of lung cancer cells through downregulation of inhibitor of apoptosis proteins and the Akt signaling pathway. Oncol Rep 2014;32(3):1087-93. https://doi.org/10.3892/or.2014.3273.
Li LQ, Li XL, Wang L, Du WJ, Guo R, Liang HH, et al. Matrine inhibits breast cancer growth via miR-21/PTEN/Akt pathway in MCF-7 cells. Cell Physiol Biochem 2012;30(3):631-41. https://doi.org/10.1159/000341444.
Zhang P, Wang Z, Chong T, Ji Z. Matrine inhibits proliferation and induces apoptosis of the androgen-independent prostate cancer cell line PC-3. Mol Med Rep 2012;5(3):783-7. https://doi.org/10.3892/mmr.2011.701.
Liu T, Song Y, Chen H, Pan S, Sun X. Matrine inhibits proliferation and induces apoptosis of pancreatic cancer cells in vitro and in vivo. Biol Pharm Bull 2010;33(10):1740-5. https://doi.org/10.1248/bpb.33.1740.
Chang C, Liu SP, Fang CH, He RS, Wang Z, Zhu YQ, et al. Effects of matrine on the proliferation of HT29 human colon cancer cells and its antitumor mechanism. Oncol Lett 2013;6(3):699-704. https://doi.org/10.3892/ol.2013.1449.
Korkmaz G, le Sage C, Tekirdag KA, Agami R, Gozuacik D. MiR-376b controls starvation and mTOR inhibition-related autophagy by targeting ATG4C and BECN1. Autophagy 2012;8(2):165-76. https://doi.org/10.4161/auto.8.2.18351.
Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S, et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci U S A 2004;101(9):2999-3004. https://doi.org/10.1073/pnas.0307323101.
Croce CM. Causes and consequences of microRNA dysregulation in cancer. Nat Rev Genet 2009;10(10):704-14. https://doi.org/10.1038/nrg2634.
Esquela-Kerscher A, Slack FJ. Oncomirs-microRNAs with a role in cancer. Nat Rev Cancer 2006;6(4):259-69. https://doi.org/10.1038/nrc1840.
Pencheva N, Tavazoie SF. Control of metastatic progression by microRNA regulatory networks. Nat Cell Biol 2013;15(6):546-54. https://doi.org/10.1038/ncb2769.
Wang T, Zhang X, Obijuru L, Laser J, Aris V, Lee P, et al. A micro-RNA signature associated with race, tumor size, and target gene activity in human uterine leiomyomas. Genes Chromosomes Cancer 2007;46(4):336-47. https://doi.org/10.1002/gcc.20415.
Yonekawa T, Thorburn A. Autophagy and cell death. Essays Biochem 2013;55:105-17. https://doi.org/10.1042/bse0550105.
Zhang L, Volinia S, Bonome T, Calin GA, Greshock J, Yang N, et al. Genomic and epigenetic alterations deregulate microRNA expression in human epithelial ovarian cancer. Proc Natl Acad Sci U S A 2008;105(19):7004-9. https://doi.org/10.1073/pnas.0801615105.
Son JW, Kim YJ, Cho HM, Lee SY, Jang JS, Choi JE, et al. MicroRNA expression profiles in Korean non-small cell lung cancer. Tubercu Respir Dis 2009;67(5):413-21. https://doi.org/10.4046/trd.2009.67.5.413.
Nakada C, Matsuura K, Tsukamoto Y, Tanigawa M, Yoshimoto T, Narimatsu T, et al. Genome-wide microRNA expression profiling in renal cell carcinoma: Significant down-regulation of miR-141 and miR-200c. J Pathol 2008;216(4):418-27. https://doi.org/10.1002/path.2437.
Yoshitaka T, Kawai A, Miyaki S, Numoto K, Kikuta K, Ozaki T, et al. Analysis of microRNAs expressions in chondrosarcoma. J Orthop Res 2013;31(12):1992-8. https://doi.org/10.1002/jor.22457.
Lu S, Jiao H, Xu J, Zheng Y, Sun Y, Chen H. Downregulation of IL6 targeted MiR-376b may contribute to a positive IL6 feedback loop during early liver regeneration in mice. Cell Physiol Biochem 2015;37(1):233-42. https://doi.org/10.1159/000430348.
Chen D, Farwell MA, Zhang B. MicroRNA as a new player in the cell cycle. J Cell Physiol 2010;225(2):296-301. https://doi.org/10.1002/jcp.22234.
Li B, Shi XB, Nori D, Chao CK, Chen AM, Valicenti R, et al. Down-regulation of microRNA 106b is involved in p21-mediated cell cycle arrest in response to radiation in prostate cancer cells. Prostate 2011;71(6):567-74. https://doi.org/10.1002/pros.21272.
An Q, Han C, Zhou Y, Li F, Li D, Zhang X, et al. Matrine induces cell cycle arrest and apoptosis with recovery of the expression of miR-126 in the A549 non-small cell lung cancer cell line. Mol Med Rep 2016;14(5):4042-8. https://doi.org/10.3892/mmr.2016.5753.
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2018 Bosnian Journal of Basic Medical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2017-10-09
Published 2018-05-20









