Effects of single-dose neuropeptide Y on levels of hippocampal BDNF, MDA, GSH, and NO in a rat model of pentylenetetrazole-induced epileptic seizure
DOI:
https://doi.org/10.17305/bjbms.2013.2332Keywords:
epileptic seizure, neuropeptide Y, brain-derived neurotrophic factor, oxidative stress, nitrosative stressAbstract
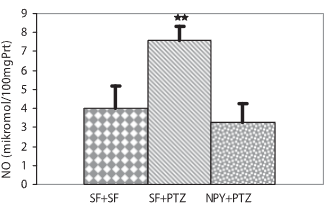
Epilepsy is one of the most common neurological disorders, characterized by recurrent seizures, which may increase the content of reactive oxygen and nitrogen species. The objective of this study was to investigate the effects of Neuropeptide Y on oxidative and nitrosative balance and brain-derived neurotrophic factor levels induced by pentylenetetrazole (a standard convulsant drug) in the hippocampus of Wistar rats. Three groups of seven rats were treated intraperitoneally as follows: group 1 (saline + saline) 1 ml saline, group 2 (salin + Pentylenetetrazole) 1 ml saline 30 min before Pentylenetetrazole; and group 3 (Neuropeptide Y + Pentylenetetrazole) 60 μg/kg Neuropeptide Y 30 min before 60 mg/kg Pentylenetetrazole. After 24 h, the animals were euthanized by decapitation. Hippocampus were isolated to evaluate the malondi-aldehyde, glutathione, nitric oxide, and brain-derived neurotrophic factor levels in three rat groups. The results of this study demonstrated that while intraperitoneally administered neuropeptide Y did not result in a statistically significant difference in BDNF levels, its administration caused a statistically significant decrease in malondialdehyde and nitric oxide levels and an increase in glutathione levels in rats with pentylenetetrazole-induced epileptic seizure. Neuropeptide Y were able to reduce nitroxidative damage induced by pentylenetetrazole in the hippocampus of Wistar rats.
Citations
Downloads
Downloads
Additional Files
Published
Issue
Section
Categories
License
Copyright (c) 2017 Bosnian Journal of Basic Medical Sciences

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2017-08-07
Published 2013-11-20









