Effect of bisoprolol on central aortic systolic pressure in Chinese hypertensive patients after the initial dose and long-term treatment
DOI:
https://doi.org/10.17305/bjbms.2021.6483Keywords:
Bisoprolol, BPro® device, central aortic systolic pressure, radial tonometryAbstract
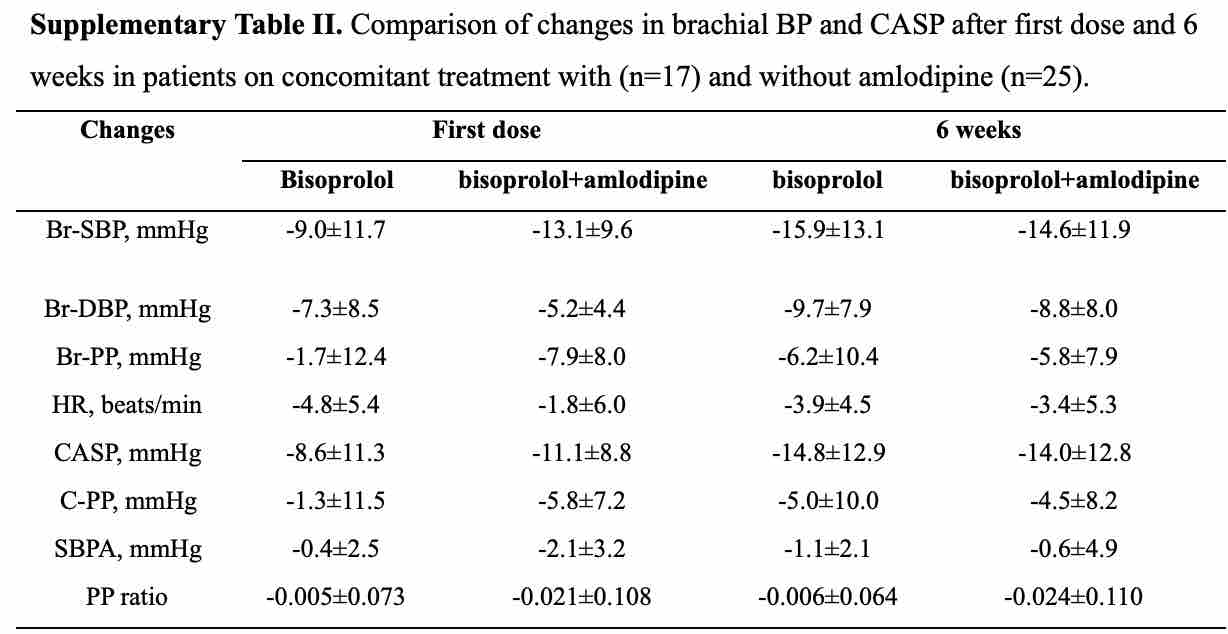
We conducted a prospective open-label cohort study with the aim of examining the effects of the highly β1-selective agent bisoprolol on central aortic systolic pressure (CASP) after the first dose and after 6 weeks’ treatment and whether the CASP response could be predicted from the early response. Chinese patients with primary hypertension (BP ≥ 140/90 mmHg) on no therapy or background amlodipine were treated with bisoprolol 2.5 mg daily for 6 weeks. Brachial systolic BP (Br-SBP), resting heart rate (HR) and CASP were determined at baseline, 24h after the first dose, and pre-dose after treatment for 6 weeks using the BPro® device. In 42 patients (age 54 ± 9 years) the mean reductions in CASP and Br-SBP after 6 weeks of treatment were not significantly different from each other at -14.5 ± 12.7 and -15.4 ± 12.9 mmHg (both p<0.01), respectively. Changes in CASP and Br-SBP were highly correlated after the first dose (r = 0.964, p<0.01) and after 6 weeks (r = 0.963, p<0.01) and the reductions in CASP after 6 weeks were also associated with the reduction in CASP after the first dose (r = 0.577, p<0.01). Bisoprolol was shown to effectively reduce CASP and this effect was directly proportional to the reduction in Br-SBP and of a similar magnitude. More favourable CASP responses to long term therapy may be predicted by greater reductions in CASP after the first dose.
Citations
Downloads
Downloads
Additional Files
Published
License
Copyright (c) 2021 Weiwei Zeng, Brian Tomlinson

This work is licensed under a Creative Commons Attribution 4.0 International License.
How to Cite
Accepted 2021-10-21
Published 2022-06-01









