Secreted frizzled-related protein 4 (sFRP4) in cancer – Dual roles in tumorigenesis and therapeutic potential: A review
DOI:
https://doi.org/10.17305/bb.2025.13047Keywords:
sFRP4, tumorigenesis, dual role, oncotherapyAbstract
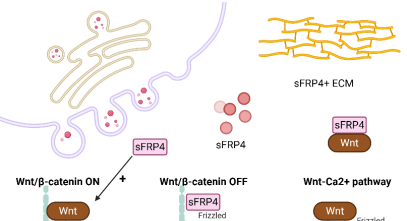
Secreted Frizzled-Related Protein 4 (sFRP4), the largest member of the Secreted Frizzled-Related Protein (sFRP) family, contains two functional domains: a cysteine-rich domain (CRD) homologous to the Wnt-binding region of Frizzled (FZD) receptors and a netrin-like (NTR) domain structurally similar to axonal guidance proteins. By modulating the Wingless/Integrated (Wnt) signaling pathway, sFRP4 regulates essential cellular processes including proliferation, differentiation, apoptosis, and tissue homeostasis. This review aims to provide a comprehensive overview of the dualistic roles of sFRP4 in cancer, highlighting its tumor-suppressive and tumor-promoting functions, underlying molecular mechanisms, and therapeutic potential. A systematic literature search was conducted in PubMed and Web of Science databases (1996–2025) using predefined keywords, and from 277 identified publications, 47 studies were included that comprised clinical data, in vitro cell models, and in vivo experimental systems. Findings demonstrate that sFRP4 frequently acts as a tumor suppressor by sequestering Wnt ligands, suppressing cancer stem cell-like properties, reprogramming tumor metabolism, inhibiting angiogenesis, and enhancing chemosensitivity. Its downregulation is often driven by promoter hypermethylation or repression mediated by microRNAs (miRNAs). Conversely, in gastrointestinal and prostate cancers, sFRP4 is frequently upregulated, where it promotes Wnt pathway activation, invasion, stemness, chemoresistance, and reshaping of the tumor immune microenvironment. Mechanistic insights indicate that post-translational modifications and nuclear localization of sFRP4 further contribute to its paradoxical context-dependent functions. In conclusion, sFRP4 exerts dual roles in tumorigenesis, acting either as a tumor suppressor or promoter depending on tissue type, tumor microenvironment, and regulatory mechanisms. This complexity underscores both the challenges and opportunities of targeting sFRP4 in oncology, and future therapeutic strategies incorporating recombinant proteins, synthetic peptides, and nanoparticle-based delivery systems hold promise for harnessing its anti-tumor potential while overcoming resistance mechanisms.
Citations
Downloads
Downloads
Published
Issue
Section
Categories
License
Copyright (c) 2025 Yu Jiang, Luyao Wang, Yerong Li, Juan Liu, Juan Lv, Pengfei Xu

This work is licensed under a Creative Commons Attribution 4.0 International License.









